A) EtOH,H+
B) CH3CH2NH2,DCC
C) SOCl2
D) Heat
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about amides is true?
A) Amides react with methyl alcohol in the presence of an acid catalyst to form an ester.
B) Amides are hydrolyzed in acid or base to form carboxylic acids or carboxylate anions.
C) Amides react with thionyl chloride to form the acid chloride.
D) Amides do not react under any conditions.They are inert compounds.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What reagents are necessary to perform the following reaction? 
A) EtOH,H+
B) CH3CH2NH2,DCC
C) SOCl2
D) Heat
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 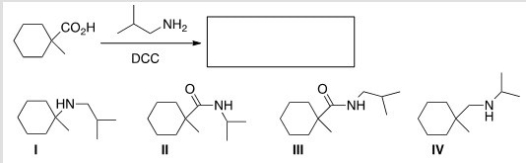
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the IUPAC name for the following compound. 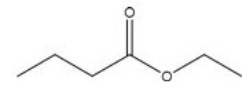
A) Butyl ethanoate
B) Ethyl butanoate
C) 4-hexanoate
D) 3-hexanoate
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the first step in the general mechanism for nucleophilic acyl substitution?
A) Protonation of the carbonyl
B) Removal of an a-proton
C) Addition of the nucleophile to the carbonyl
D) Loss of the leaving group
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the direct product of the base-promoted hydrolysis of an ester?
A) A nitrile
B) A carboxylic acid
C) An amide
D) A carboxylic acid salt
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why doesn't nucleophilic acyl substitution stop at the tetrahedral intermediate?
A) The nucleophile is too basic.
B) Reforming the carbonyl is energetically favorable.
C) The leaving group is unstable and wants to be negatively charged.
D) There is no tetrahedral intermediate.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is an amide less reactive to nucleophilic acyl substitution than an acid chloride?
A) Nitrogen is a better leaving group.
B) Chloride is a better leaving group.
C) Nitrogen donates more electron density into the carbonyl.
D) The amide anion is less basic.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Where do the carbonyl signals appear in the 13C NMR spectrum of carboxylic acid derivatives?
A) 1700 cm-1
B) 180-160 ppm
C) 2) 5-3.0 ppm
D) 100-80 ppm
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the IUPAC name for the following compound. 
A) R-3-methoxybutanoyl chloride
B) S-3-methoxybutanoyl chloride
C) R-2-methoxy-4-butanoyl chloride
D) S-2-methoxy-4-butanoyl chloride
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Where is the carbonyl absorption found in the IR spectrum of a simple ester?
A) 2) 5 ppm
B) 2250 cm-1
C) 3800 cm-1
D) 1740 cm-1
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
You see an absorption at 2250 cm-1 in the IR spectrum of a compound.What kind of functional group is present?
A) A ketone
B) An aldehyde
C) An ester
D) A nitrile
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of increasing reactivity in nucleophilic acyl substitution. 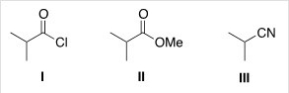
A) I < II < III
B) III < II < I
C) II < III < I
D) III < I < II
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 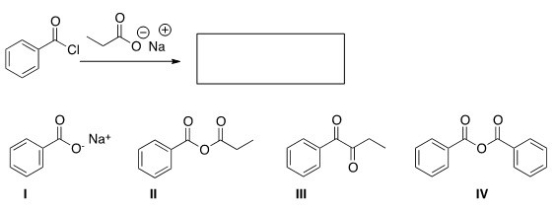
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the missing reagent in the reaction below? 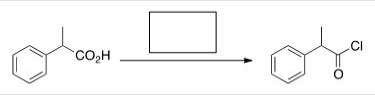
A) Acetic acid
B) NaOMe
C) SOCl2
D) Pyridine
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a lactam? 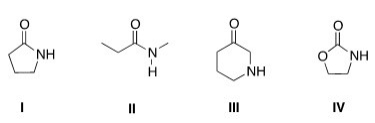
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Will the following reaction occur? 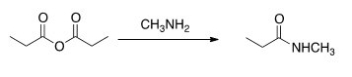
A) Yes
B) No
D) undefined
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 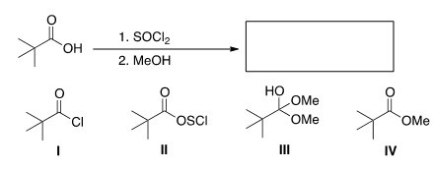
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name for the following compound? 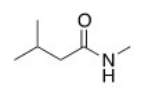
A) Methyl 3-methylbutanamide
B) 5-Methylhexanamide
C) N-Methyl 3-methylhexanamide
D) N-Methyl 3-methylbutanamide
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 50
Related Exams