A) 214 g
B) 245 g
C) 321 g
D) 489 g
E) 643 g
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Aluminum oxide, Al2O3, is used as a filler for paints and varnishes as well as in the manufacture of electrical insulators. Calculate the number of moles in 47.51 g of Al2O3.
A) 2.377 mol
B) 2.146 mol
C) 1.105 mol
D) 0.4660 mol
E) 0.4207 mol
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
One mole of methane (CH4) contains a total of 3 × 1024 atoms.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Balance the following equation: UO2(s) + HF(l) → UF4(s) + H2O(l)
A) UO2(s) + 2HF(l) → UF4(s) + H2O(l)
B) UO2(s) + 4HF(l) → UF4(s) + 2H2O(l)
C) UO2(s) + H4F4(l) → UF4 (s) + H4O2(l)
D) UO2(s) + 4HF(l) → UF4(s) + 4H2O(l)
E) UO2(s) + 8HF(l) → 2UF4(s) + 4H2O(l)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Household sugar, sucrose, has the molecular formula C12H22O11. What is the percent of carbon in sucrose, by mass?
A) 26.7%
B) 33.3%
C) 41.4%
D) 42.1%
E) 52.8%
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following samples has the most moles of the compound?
A) 50.0 g of Li2O
B) 75.0 g of CaO
C) 200.0 g of Fe2O3
D) 50.0 g of CO2
E) 100.0 g of SO3
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Aluminum oxide (used as an adsorbent or a catalyst for organic reactions) forms when aluminum reacts with oxygen. 4Al(s) + 3O2(g) → 2Al2O3(s)
A mixture of 82.49 g of aluminum ( 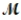 = 26.98 g/mol) and 117.65 g of oxygen (
= 26.98 g/mol) and 117.65 g of oxygen ( 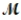 = 32.00g/mol) is allowed to react. What mass of aluminum oxide (
= 32.00g/mol) is allowed to react. What mass of aluminum oxide ( 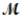 = 101.96 g/mol) can be formed?
= 101.96 g/mol) can be formed?
A) 155.8 g
B) 200.2 g
C) 249.9 g
D) 311.7 g
E) 374.9 g
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the percent yield for the reaction PCl3(g) + Cl2(g) → PCl5(g)
If 119.3 g of PCl5 (M = 208.2 g/mol) are formed when 61.3 g of Cl2 ( 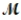 = 70.91 g/mol) react with excess PCl3?
= 70.91 g/mol) react with excess PCl3?
A) 195%
B) 85.0%
C) 66.3%
D) 51.4%
E) 43.7%
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Balance the following equation: C8H18O3(l) + O2(g) → H2O(g) + CO2(g)
A) C8H18O3(l) + 8O2(g) → 9H2O(g) + 8CO2(g)
B) C8H18O3(l) + 11O2(g) → 9H2O(g) + 8CO2(g)
C) 2C8H18O3(l) + 22O2(g) → 9H2O(g) + 16CO2(g)
D) C8H18O3(l) + 13O2(g) → 18H2O(g) + 8CO2(g)
E) 2C8H18O3(l) + 17O2(g) → 18H2O(g) + 16CO2(g)
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Phosphine, an extremely poisonous and highly reactive gas, will react with oxygen to form tetraphosphorus decaoxide and water. PH3(g) + O2(g) → P4O10(s) + H2O(g) [unbalanced] Calculate the mass of P4O10(s) formed when 225 g of PH3 reacts with excess oxygen.
A) 1880 g
B) 940. g
C) 900. g
D) 470 g
E) 56.3 g
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Potassium chloride is used as a substitute for sodium chloride for individuals with high blood pressure. Identify the limiting reactant and determine the mass of the excess reactant remaining when 7.00 g of chlorine gas reacts with 5.00 g of potassium to form potassium chloride.
A) Potassium is the limiting reactant; 2.47 g of chlorine remain.
B) Potassium is the limiting reactant; 7.23 g of chlorine remain.
C) Chlorine is the limiting reactant; 4.64 g of potassium remain.
D) Chlorine is the limiting reactant; 2.70 g of potassium remain.
E) No limiting reagent: the reactants are present in the correct stoichiometric ratio.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Analysis of a carbohydrate showed that it consisted of 40.0 % C, 6.71 % H, and 53.3 % O by mass. Its molecular mass was found to be between 140 and 160 amu. What is the molecular formula of this compound?
A) C4H8O6
B) C5H10O5
C) C5H12O5
D) C6H12O4
E) None of these choices are correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Phosphorus pentachloride, PCl5, a white solid that has a pungent, unpleasant odor, is used as a catalyst for certain organic reactions. Calculate the number of moles in 38.7 g of PCl5.
A) 5.38 mol
B) 3.55 mol
C) 0.583 mol
D) 0.282 mol
E) 0.186 mol
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Balance the following equation: B2O3(s) + HF(l) → BF3(g) + H2O(l)
A) B2O3(s) + 6HF(l) → 2BF3(g) + 3H2O(l)
B) B2O3 (s) + H6F6(l) → B2F6(g) + H6O3(l)
C) B2O3 (s) + 2HF(l) → 2BF3(g) + H2O(l)
D) B2O3 (s) + 3HF(l) → 2BF3(g) + 3H2O(l)
E) B2O3 (s) + 6HF(l) → 2BF3(g) + 6H2O(l)
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sodium bromate is used in a mixture which dissolves gold from its ores. Calculate the mass in grams of 4.68 mol of sodium bromate.
A) 706 g
B) 482 g
C) 383 g
D) 32.2 g
E) 0.0310 g
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ammonia, an important source of fixed nitrogen that can be metabolized by plants, is produced using the Haber process in which nitrogen and hydrogen combine. N2(g) + 3H2(g) → 2NH3(g) How many grams of nitrogen are needed to produce 325 grams of ammonia?
A) 1070 g
B) 535 g
C) 267 g
D) 178 g
E) 108 g
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Tetraphosphorus hexaoxide ( 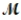 = 219.9 g/mol) is formed by the reaction of phosphorus with oxygen gas. P4(s) + 3O2(g) → P4O6(s)
If a mixture of 75.3 g of phosphorus and 38.7 g of oxygen produce 43.3 g of P4O6, what is the percent yield for the reaction?
= 219.9 g/mol) is formed by the reaction of phosphorus with oxygen gas. P4(s) + 3O2(g) → P4O6(s)
If a mixture of 75.3 g of phosphorus and 38.7 g of oxygen produce 43.3 g of P4O6, what is the percent yield for the reaction?
A) 57.5%
B) 48.8%
C) 38.0%
D) 32.4%
E) 16.3%
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A compound of bromine and fluorine is used to make UF6, which is an important chemical in processing and reprocessing of nuclear fuel. The compound contains 58.37 mass percent bromine. Determine its empirical formula.
A) BrF
B) BrF2
C) Br2F3
D) Br3F
E) BrF3
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sulfur dioxide reacts with chlorine to produce thionyl chloride (used as a drying agent for inorganic halides) and dichlorine oxide (used as a bleach for wood, pulp, and textiles) . SO2(g) + 2Cl2(g) → SOCl2(g) + Cl2O(g) If 0.400 mol of Cl2 reacts with excess SO2, how many moles of Cl2O are formed?
A) 0.800 mol
B) 0.400 mol
C) 0.200 mol
D) 0.100 mol
E) 0.0500 mol
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A normal breath takes in about 1.0 L of air. Assuming that air has an average molar mass of 28.8g, and that its density is 0.97 g/L, how many molecules of air do you take in with each breath?
A) 2.0 × 1022
B) 2.2 × 1022
C) 5.8 × 1023
D) 1.7 × 1025
E) 1.8 × 1025
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 70
Related Exams