A) 118.15 g/mol
B) 99.15 g/mol
C) 78.07 g/mol
D) 59.08 g/mol
E) 50.01 g/mol
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Aluminum reacts with oxygen to produce aluminum oxide which can be used as an adsorbent, desiccant, or catalyst for organic reactions. 4Al(s) + 3O2(g) → 2Al2O3(s)
A mixture of 82.49 g of aluminum ( 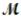 = 26.98 g/mol) and 117.65 g of oxygen (
= 26.98 g/mol) and 117.65 g of oxygen ( 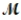 = 32.00 g/mol) is allowed to react. Identify the limiting reactant and determine the mass of the excess reactant present in the vessel when the reaction is complete.
= 32.00 g/mol) is allowed to react. Identify the limiting reactant and determine the mass of the excess reactant present in the vessel when the reaction is complete.
A) Oxygen is the limiting reactant; 19.81 g of aluminum remain.
B) Oxygen is the limiting reactant; 35.16 g of aluminum remain.
C) Aluminum is the limiting reactant; 16.70 g of oxygen remain.
D) Aluminum is the limiting reactant; 35.16 g of oxygen remain.
E) Aluminum is the limiting reactant; 44.24 g of oxygen remain.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Gadolinium oxide, a colorless powder which absorbs carbon dioxide from the air, contains 86.76 mass % Gd. Determine its empirical formula.
A) Gd2O3
B) Gd3O2
C) Gd3O4
D) Gd4O3
E) GdO
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Potassium chlorate (used in fireworks, flares, and safety matches) forms oxygen and potassium chloride when heated. KClO3(s) → KCl(s) + O2(g) [unbalanced] How many grams of oxygen are formed when 26.4 g of potassium chlorate is heated?
A) 223 g
B) 99.1 g
C) 10.3 g
D) 6.86 g
E) 4.60 g
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In a blast furnace, elemental iron is produced from a mixture of coke (C) , iron ore (Fe3O4) , and other reactants. An important reaction sequence is 2C(s) + O2(g) → 2CO(g) Fe3O4(s) + 4CO(g) → 3Fe(l) + 4CO2(g) How many moles of iron can be formed in this sequence when 1.00 mol of carbon, as coke, is consumed?
A) 6.00 mol Fe
B) 3.00 mol Fe
C) 1.33 mol Fe
D) 1.25 mol Fe
E) 0.750 mol Fe
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hydroxylamine nitrate contains 29.17 mass % N, 4.20 mass % H, and 66.63 mass % O. Determine its empirical formula.
A) HNO
B) H2NO2
C) HN6O16
D) HN16O7
E) H2NO3
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the molar mass of Ca(BO2) 2·6H2O.
A) 273.87 g/mol
B) 233.79 g/mol
C) 183.79 g/mol
D) 174.89 g/mol
E) 143.71 g/mol
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Copper(II) sulfate pentahydrate, CuSO4·5H2O, is used as a fungicide and algicide. Calculate the mass of oxygen in 1.000 mol of CuSO4·5H2O.
A) 249.7 g
B) 144.0 g
C) 96.00 g
D) 80.00 g
E) 64.00 g
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many grams of sodium fluoride (used in water fluoridation and manufacture of insecticides) are needed to form 485 g of sulfur tetrafluoride? 3SCl2(l) + 4NaF(s) → SF4(g) + S2Cl2(l) + 4NaCl(s)
A) 1940 g
B) 1510 g
C) 754 g
D) 205 g
E) 51.3 g
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many grams of oxygen are needed to react completely with 200.0 g of ammonia, NH3? 4NH3(g) + 5O2(g) → 4NO(g) + 6H2O(g)
A) 469.7 g
B) 300.6 g
C) 250.0 g
D) 3.406 g
E) 2.180 g
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Balance the following equation for the combustion of benzene: C6H6(l) + O2(g) → H2O(g) + CO2(g)
A) C6H6(l) + 9O2(g) → 3H2O(g) + 6CO2(g)
B) C6H6(l) + 9O2(g) → 6H2O(g) + 6CO2(g)
C) 2C6H6(l) + 15O2(g) → 6H2O(g) + 12CO2(g)
D) C6H6(l) + 15O2(g) → 3H2O(g) + 6CO2(g)
E) 2C6H6(l) + 9O2(g) → 6H2O(g) + 12CO2(g)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An important reaction sequence in the industrial production of nitric acid is the following: N2(g) + 3H2(g) → 2NH3(g) 4NH3(g) + 5O2(g) → 4NO(g) + 6H2O(l) Starting from 20.0 mol of nitrogen gas in the first reaction, how many moles of oxygen gas are required in the second one?
A) 12.5 mol O2
B) 20.0 mol O2
C) 25.0 mol O2
D) 50.0 mol O2
E) 100. mol O2
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Lead(II) sulfide was once used in glazing earthenware. It will also react with hydrogen peroxide to form lead(II) sulfate and water. How many grams of hydrogen peroxide are needed to react completely with 265 g of lead(II) sulfide?
A) 151 g
B) 123 g
C) 50.3 g
D) 37.7 g
E) 9.41 g
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Potassium dichromate, K2Cr2O7, is used in tanning leather, decorating porcelain, and water proofing fabrics. Calculate the number of chromium atoms in 78.82 g of K2Cr2O7.
A) 9.490 × 1025 Cr atoms
B) 2.248 × 1024 Cr atoms
C) 1.124 × 1024 Cr atoms
D) 3.227 × 1023 Cr atoms
E) 1.613 × 1023 Cr atoms
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The formula CH3O0.5 is an example of an empirical formula.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Magnesium fluoride is used in the ceramics and glass industry. What is the mass of 1.72 mol of magnesium fluoride?
A) 43.3 g
B) 62.3 g
C) 74.5 g
D) 92.9 g
E) 107 g
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Lead (II) nitrate is a poisonous substance which has been used in the manufacture of special explosives and as a sensitizer in photography. Calculate the mass of lead in 139 g of Pb(NO3) 2.
A) 107 g
B) 90.8 g
C) 87.0 g
D) 83.4 g
E) 62.6 g
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
In a correctly balanced equation, the number of reactant molecules must equal the number of product molecules.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the percent composition of potassium dichromate, K2Cr2O7.
A) 17.5% K, 46.6% Cr, 35.9% O
B) 29.8% K, 39.7% Cr, 30.5% O
C) 36.5% K, 48.6% Cr, 14.9% O
D) 37.2% K, 24.7% Cr, 38.1% O
E) None of these choices are correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the combustion analysis of 0.1127 g of glucose (C6H12O6) , what mass, in grams, of CO2 would be produced?
A) 0.0451 g
B) 0.0825 g
C) 0.1652 g
D) 0.4132 g
E) 1.466 g
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 70
Related Exams