B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following combinations of names and formulas of ions is incorrect?
A) Ba2+ barium
B) S2- sulfate
C) CN- cyanide
D) ClO4- perchlorate
E) HCO3- bicarbonate
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When an atom is represented by the symbol 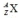 , the value of A is the
, the value of A is the
A) number of neutrons in the atom.
B) number of protons in the atom.
C) atomic mass of the element.
D) total number of electrons and neutrons in the atom.
E) total number of protons and neutrons in the atom.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the molecular mass of iron (III) bromide hexahydrate, a substance used as a catalyst in organic reactions.
A) 403.65 amu
B) 355.54 amu
C) 317.61 amu
D) 313.57 amu
E) 295.56 amu
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a metalloid?
A) carbon, C, Z = 6
B) sulfur, S, Z = 16
C) germanium, Ge, Z = 32
D) iridium, Z = 77
E) bromine, Br, Z = 35
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the empirical formula for hexane, C6H14?
A) C12H28
B) C6H14
C) C3H7
D) CH2.3
E) C0.43H
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Lithium forms compounds which are used in dry cells and storage batteries and in high-temperature lubricants. It has two naturally occurring isotopes, 6Li (isotopic mass = 6.015121 amu) and 7Li (isotopic mass = 7.016003 amu) . Lithium has an atomic mass of 6.9409 amu. What is the percent abundance of lithium-6?
A) 92.50%
B) 86.66%
C) 46.16%
D) 7.503%
E) 6.080%
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ferric oxide is used as a pigment in metal polishing. Which of the following is its formula?
A) FeO
B) Fe2O
C) FeO3
D) Fe2O5
E) Fe2O3
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Modern studies have shown that the Law of Multiple Proportions is not valid.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The substance, CoCl2, is useful as a humidity indicator because it changes from pale blue to pink as it gains water from moist air. What is its name?
A) cobalt dichloride
B) cobalt(II) chloride
C) cobalt chloride
D) cobaltic chloride
E) copper(II) chloride
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A row of the periodic table is called a
A) group.
B) period.
C) isotopic mixture.
D) family.
E) subshell.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name of PCl3?
A) phosphorus chloride
B) phosphoric chloride
C) phosphorus trichlorate
D) trichlorophosphide
E) phosphorus trichloride
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
In nature, some elements exist as molecules, while others do not.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following combinations of names and formulas of ions is incorrect?
A) O2- oxide
B) Cd2+ cadmium
C) ClO3- chlorate
D) HCO3- hydrogen carbonate
E) NO2- nitrate
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A red glaze on porcelain can be produced by using MnSO4. What is its name?
A) manganese disulfate
B) manganese(II) sulfate
C) manganese(IV) sulfate
D) manganese sulfate
E) manganese(I) sulfate
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Barium sulfate is used in manufacturing photographic paper. What is its formula?
A) BaSO4
B) Ba(SO4) 2
C) Ba2SO4
D) Ba2(SO4) 3
E) BaSO3
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The substance, KClO3, is a strong oxidizer used in explosives, fireworks, and matches. What is its name?
A) potassium chlorite
B) potassium chloride
C) potassium(I) chlorite
D) potassium(I) chlorate
E) potassium chlorate
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In a Millikan oil-drop experiment, the charges on several different oil drops were as follows: -5.92; -4.44; -2.96; -8.88. The units are arbitrary. What is the likely value of the electronic charge in these arbitrary units?
A) -1.11
B) -1.48
C) -2.22
D) -2.96
E) -5.55
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ions occurs commonly?
A) N3+
B) S6+
C) O2-
D) Ca+
E) Cl+
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name of BBr3?
A) boron bromide
B) boric bromide
C) boron tribromide
D) tribromoboride
E) bromine triboride
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 100
Related Exams