A) 2.97 × 10-4
B) 1.72 × 10-2
C) 3.45 × 10-2
D) 1.31 × 10-1
E) > 1.00
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
H2SO3(aq)  HSO3(aq) + H+(aq) Kc = 1.4 × 10-2 H2SO3(aq)
HSO3(aq) + H+(aq) Kc = 1.4 × 10-2 H2SO3(aq)  SO32-(aq) + 2H+(aq) Kc = 9.1 × 10-10
Given the above equilibrium constant data at 25°C, what is the value of Kc at this temperature for the reaction
HSO3-(aq)
SO32-(aq) + 2H+(aq) Kc = 9.1 × 10-10
Given the above equilibrium constant data at 25°C, what is the value of Kc at this temperature for the reaction
HSO3-(aq)  SO32-(aq) + H+(aq) ?
SO32-(aq) + H+(aq) ?
A) 6.5 ×10-8
B) 1.3 × 10-11
C) 7.8 × 1010
D) 1.5 × 107
E) None of these choices are correct.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sodium hydrogen carbonate decomposes above 110°C to form sodium carbonate, water, and carbon dioxide. 2NaHCO3(s)  Na2CO3(s) + H2O(g) + CO2(g)
One thousand grams of sodium hydrogen carbonate are added to a reaction vessel, the temperature is increased to 200°C, and the system comes to equilibrium. What happens in this system if another 50 g of sodium carbonate are now added?
Na2CO3(s) + H2O(g) + CO2(g)
One thousand grams of sodium hydrogen carbonate are added to a reaction vessel, the temperature is increased to 200°C, and the system comes to equilibrium. What happens in this system if another 50 g of sodium carbonate are now added?
A) The partial pressure of carbon dioxide will increase.
B) The partial pressure of carbon dioxide will decrease.
C) The partial pressure of carbon dioxide will be unchanged.
D) The amounts of all products will be greater when equilibrium is reestablished.
E) None of these choices are correct.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following has an effect on the magnitude of the equilibrium constant?
A) removing products as they are formed
B) adding more of a reactant
C) adding a catalyst
D) increasing the pressure, in a gas-phase reaction
E) change in temperature
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following two equilibria and their respective equilibrium constants: (1) NO(g) + ½O2(g)  NO2(g)
(2) 2NO2(g)
NO2(g)
(2) 2NO2(g)  2NO(g) + O2(g)
Which one of the following is the correct relationship between the equilibrium constants K1 and K2?
2NO(g) + O2(g)
Which one of the following is the correct relationship between the equilibrium constants K1 and K2?
A) K2 = 2/K1
B) K2 = (1/K1) 2
C) K2 = -K1/2
D) K2 = 1/(2K1)
E) K2 = 1/(2K1) 2
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the mass-action expression,  c, for the following chemical reaction? Cu2+(aq) + 4NH3(aq)
c, for the following chemical reaction? Cu2+(aq) + 4NH3(aq)  Cu(NH3) 42+(aq)
Cu(NH3) 42+(aq)
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) None of these choices are correct.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
10.0 mL of a 0.100 mol L-1 solution of a metal ion M2+ is mixed with 10.0 mL of a 0.100 mol L-1 solution of a substance L. The following equilibrium is established: M2+(aq) + 2L(aq)  ML22+(aq)
At equilibrium the concentration of L is found to be 0.0100 mol L-1. What is the equilibrium concentration of ML22+, in mol L-1?
ML22+(aq)
At equilibrium the concentration of L is found to be 0.0100 mol L-1. What is the equilibrium concentration of ML22+, in mol L-1?
A) 0.100 mol L-1
B) 0.050 mol L-1
C) 0.025 mol L-1
D) 0.0200 mol L-1
E) 0.0100 mol L-1
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A chemical reaction has an equilibrium constant of 2 × 106. If this reaction is at equilibrium, select the one correct conclusion that can be made about the reaction.
A) The forward and back reactions have stopped.
B) The limiting reactant has been used up.
C) The forward and reverse rate constants are equal.
D) The forward and reverse reaction rates are equal.
E) None of these choices are correct.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Increasing the initial amount of the limiting reactant in a reaction will increase the value of the equilibrium constant, Kc.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
In a chemical reaction, if the starting concentrations of reactants are increased, then the equilibrium constant Kc will also increase.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Nitric oxide and bromine were allowed to react in a sealed container. When equilibrium was reached PNO = 0.526 atm, 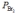 = 1.59 atm, and PNOBr = 7.68 atm. Calculate Kp for the reaction.
2NO(g) + Br2(g)
= 1.59 atm, and PNOBr = 7.68 atm. Calculate Kp for the reaction.
2NO(g) + Br2(g)  2NOBr(g)
2NOBr(g)
A) 7.45 × 10-3
B) 0.109
C) 9.18
D) 91.8
E) 134
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The equilibrium constant for reaction (1) below is 276. Under the same conditions, what is the equilibrium constant of reaction (2) ? (1) ½X2(g) + ½Y2(g)  XY(g)
(2) 2XY(g)
XY(g)
(2) 2XY(g)  X2(g) + Y2(g)
X2(g) + Y2(g)
A) 6.02 × 10-2
B) 7.25 × 10-3
C) 3.62 × 10-3
D) 1.31 × 10-5
E) None of these choices are correct.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
If all of the coefficients in the balanced equation for an equilibrium reaction are doubled, then the value of the equilibrium constant, Kc, will also be doubled.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Magnesium hydroxide is used in several antacid formulations. When it is added to water it dissociates into magnesium and hydroxide ions. Mg(OH) 2(s)  Mg2+(aq) + 2OH-(aq)
The equilibrium constant at 25°C is 8.9 × 10-12. One hundred grams of magnesium hydroxide is added to 1.00 L of water and equilibrium is established. What happens to the solution if another 10 grams of Mg(OH) 2 are now added to the mixture?
Mg2+(aq) + 2OH-(aq)
The equilibrium constant at 25°C is 8.9 × 10-12. One hundred grams of magnesium hydroxide is added to 1.00 L of water and equilibrium is established. What happens to the solution if another 10 grams of Mg(OH) 2 are now added to the mixture?
A) The hydroxide ion concentration will decrease.
B) The hydroxide ion concentration will increase.
C) The hydroxide ion concentration will be unchanged.
D) The solution will become supersaturated.
E) None of these choices are correct.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Methanol can be synthesized by combining carbon monoxide and hydrogen. CO(g) + 2H2(g)  CH3OH(g)
A reaction vessel contains the three gases at equilibrium with a total pressure of 1.00 atm. What will happen to the partial pressure of hydrogen if enough argon is added to raise the total pressure to 1.4 atm?
CH3OH(g)
A reaction vessel contains the three gases at equilibrium with a total pressure of 1.00 atm. What will happen to the partial pressure of hydrogen if enough argon is added to raise the total pressure to 1.4 atm?
A) The partial pressure of hydrogen will decrease.
B) The partial pressure of hydrogen will increase.
C) The partial pressure of hydrogen will be unchanged.
D) Kp needs to be known before a prediction can be made.
E) Both Kp and the temperature need to be known before a prediction can be made.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
Chemical reactions generally reach equilibrium because one of the reactants is used up.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following reaction is at equilibrium in a closed container. CuSO4.5H2O(s)  CuSO4(s) + 5H2O(g)
Which, if any, of the following actions will lead to an increase in the pressure of H2O present at equilibrium?
CuSO4(s) + 5H2O(g)
Which, if any, of the following actions will lead to an increase in the pressure of H2O present at equilibrium?
A) increasing the volume of the container
B) decreasing the volume of the container
C) adding a catalyst
D) removing some solid CuSO4
E) None of these choices are correct.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A mixture of 0.500 mole of carbon monoxide and 0.400 mole of bromine was placed into a rigid 1.00-L container and the system was allowed to come to equilibrium. The equilibrium
Concentration of COBr2 was 0.233 M. What is the value of Kc for this reaction?
CO(g) + Br2(g)  COBr2(g)
COBr2(g)
A) 5.23
B) 1.22
C) 1.165
D) 0.858
E) 0.191
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ammonium iodide dissociates reversibly to ammonia and hydrogen iodide. NH4I(s)  NH3(g) + HI(g)
At 400°C, Kp = 0.215. Calculate the partial pressure of ammonia at equilibrium when a sufficient quantity of ammonium iodide is heated to 400°C.
NH3(g) + HI(g)
At 400°C, Kp = 0.215. Calculate the partial pressure of ammonia at equilibrium when a sufficient quantity of ammonium iodide is heated to 400°C.
A) 0.103 atm
B) 0.215 atm
C) 0.232 atm
D) 0.464 atm
E) 2.00 atm
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Write the mass-action expression,  c, for the following chemical reaction equation.
2C6H6(g) + 15O2(g)
c, for the following chemical reaction equation.
2C6H6(g) + 15O2(g)  12CO2(g) + 6H2O(g)
12CO2(g) + 6H2O(g)
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 97
Related Exams