A) 3.3°C
B) 1.1°C
C) 0.0°C
D) -1.1°C
E) -3.3°C
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Potassium hydrogen phosphate is used in the preparation of nondairy powdered creamers. Calculate the molarity of a solution prepared by dissolving 238 g of K2HPO4 in enough water to produce 275 mL of solution.
A) 0.732 M
B) 0.865 M
C) 2.66 M
D) 4.97 M
E) None of these choices are correct.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 0.89% (w/v) sodium chloride solution is referred to as physiological saline solution because it has the same concentration of salts as human blood. What is the molarity of a physiological saline solution?
A) 0.0028 M
B) 0.015 M
C) 0.15 M
D) 0.30 M
E) 0.35 M
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
From the following list of aqueous solutions and water, select the one with the lowest freezing point.
A) 0.75 m (NH4) 3PO4
B) 1.0 m CaSO4
C) 1.0 m LiClO4
D) 1.5 m CH3OH, methyl alcohol
E) pure water
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hexachlorophene is used as a disinfectant in germicidal soaps. What mass of hexachlorophene ( 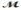 = 406.9 g/mol) must be added to 125 g of chloroform to give a solution with a boiling point of 62.60°C? Kb = 3.63°C/m, boiling point of pure chloroform = 61.70°C
= 406.9 g/mol) must be added to 125 g of chloroform to give a solution with a boiling point of 62.60°C? Kb = 3.63°C/m, boiling point of pure chloroform = 61.70°C
A) 12.6 g
B) 17.2 g
C) 31.0 g
D) 34.4 g
E) 101 g
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A solution of ethanol (C2H6O) in water is sometimes used as a disinfectant. 1.00 L of this solution contains 553 g of ethanol and 335 g of water. What is the mole fraction of ethanol in this solution?
A) 0.377
B) 0.392
C) 0.553
D) 0.608
E) 0.623
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The backbone of a protein chain consists of carbon, nitrogen, and oxygen atoms.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Barbiturates are synthetic drugs used as sedatives and hypnotics. Barbital ( 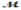 = 184.2 g/mol) is one of the simplest of these drugs. What is the boiling point of a solution prepared by dissolving 42.5 g of barbital in 825 g of acetic acid? Kb = 3.07°C/m; boiling point of pure acetic acid = 117.9°C
= 184.2 g/mol) is one of the simplest of these drugs. What is the boiling point of a solution prepared by dissolving 42.5 g of barbital in 825 g of acetic acid? Kb = 3.07°C/m; boiling point of pure acetic acid = 117.9°C
A) 117.0°C
B) 117.7°C
C) 118.1°C
D) 118.8°C
E) >120°C
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molality of a solution prepared by dissolving 86.9 g of diethyl ether, C4H10O, in 425 g of benzene, C6H6?
A) 0.362 m
B) 0.498 m
C) 2.01 m
D) 2.76 m
E) None of these choices are correct.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Carbon tetrachloride, once widely used in fire extinguishers and as a dry cleaning fluid, has been found to cause liver damage to those exposed to its vapors over long periods of time. What is the boiling point of a solution prepared by dissolving 375 g of sulfur (S8, 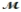 = 256.5 g/mol) in 1250 g of CCl4? Kb = 5.05°C/m, boiling point of pure CCl4 = 76.7°C?
= 256.5 g/mol) in 1250 g of CCl4? Kb = 5.05°C/m, boiling point of pure CCl4 = 76.7°C?
A) 70.8°C
B) 75.2°C
C) 78.2°C
D) 82.6°C
E) >85°C
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Under physiological conditions, amino acids in solution carry
A) no electrical charges.
B) a net positive charge, due to an -NH3+ group or groups.
C) a net negative charge, due to a -COO- group or groups.
D) both -NH2 and -COOH groups.
E) both -NH3+ and -COO- groups.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following aqueous solutions will have the lowest osmotic pressure?
A) 0.10 m KOH
B) 0.10 m RbCl
C) 0.05 m CaSO4
D) 0.05 m BaCl2
E) 0.10 m K2SO4
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the freezing point of a solution made by dissolving 3.50 g of potassium chloride ( 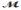 = 74.55 g/mol) in 100.0 g of water. Assume ideal behavior for the solution; Kf = 1.86°C/m.
= 74.55 g/mol) in 100.0 g of water. Assume ideal behavior for the solution; Kf = 1.86°C/m.
A) -1.7°C
B) -0.9°C
C) 0.0°C
D) 0.9°C
E) 1.7°C
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following pairs of ions is arranged so that the ion with the smaller charge density is listed first?
A) K+, Rb+
B) Cl-, K+
C) Cl-, Br-
D) Ca2+, Ba2+
E) None of these choices are correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The larger the ion, the greater in magnitude will be its heat of hydration.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The solubility of the oxidizing agent potassium permanganate is 7.1 g per 100.0 g of water at 25°C. What is the mole fraction of potassium permanganate in this solution?
A) 0.0080
B) 0.0086
C) 0.066
D) 0.45
E) 0.48
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many moles of solute particles are present in 100.0 mL of 2.50 M (NH4) 3PO4?
A) 0.100 mol
B) 0.250 mol
C) 0.500 mol
D) 0.750 mol
E) 1.00 mol
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 0.100 m MgSO4 solution has a freezing point of -0.23°C. What is the van't Hoff factor for this solution? Kf = 1.86°C/m
A) 0.62
B) 1.0
C) 1.2
D) 2.0
E) 4.0
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calcium nitrite is used as a corrosion inhibitor in lubricants. What is the molality of a solution prepared by dissolving 18.5 g of calcium nitrite in 83.5 g of distilled water?
A) 0.0342 m
B) 0.0855 m
C) 0.222 m
D) 0.444 m
E) 1.68 m
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
All solutions are mixtures.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 96
Related Exams