A) manganese, Mn, E = -1.18 V
B) cadmium, Cd, E = -0.40 V
C) magnesium, Mg, E = -2.37 V
D) zinc, Zn, E = -0.76 V
E) All of these metals are capable of acting as sacrificial anodes with iron.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
In the shorthand notation for cells, a double vertical line is used to separate the reduced and oxidized forms of a redox couple.
B) False
Correct Answer

verified
False
Correct Answer
verified
Multiple Choice
In the electrolysis of water, how many grams of oxygen gas will be produced for every gram of hydrogen gas formed? Reaction: 2H2O(l) 2H2(g) + O2(g)
A) 31.7 g
B) 15.9 g
C) 7.94 g
D) 3.97 g
E) 1.98 g
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate G for the reaction of iron(II) ions with one mole of permanganate ions. 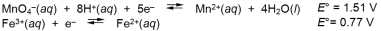
A) -71.4 kJ
B) -286 kJ
C) -357 kJ
D) -428 kJ
E) None of these choices is correct.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which component of the following cell notation is the anode? P | Q || R | S
A) P
B) Q
C) R
D) S
E) One of the | symbols is the anode.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Examine the following half-reactions and select the weakest oxidizing agent among the species listed. 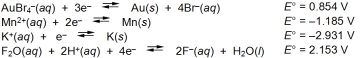
A) AuBr4¯(aq)
B) Mn2+(aq)
C) K+(aq)
D) F2O(aq)
E) H+(aq)
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
In the electrolyte of an electrochemical cell, current is carried by anions moving toward the anode and cations moving in the opposite direction.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
In the electrolyte of an electrochemical cell, current is carried by electrons moving from the anode to the cathode.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the reaction in the lead-acid cell 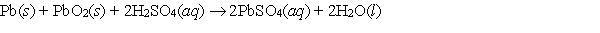 for which E cell = 2.04 V at 298 K. G for this reaction is
for which E cell = 2.04 V at 298 K. G for this reaction is
A) -3.94 * 105 kJ.
B) -3.94 * 102 kJ.
C) -1.97*105 kJ.
D) -7.87* 102 kJ.
E) None of these choices is correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following pairs of substances could be used to construct a single redox electrode (i.e., they have an element in common, but in different oxidation states) ?
A) HCl and Cl¯
B) H+ and OH¯
C) H2O and H+
D) Fe3+ and Fe2O3
E) MnO2 and Mn2+
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is not a redox reaction?
A) Al(OH) 4¯(aq) + 4H+(aq) Al3+(aq) + 4H2O(l)
B) C6H12O6(s) + 6O2(g) 6CO2(g) + 6H2O(l)
C) Na6FeCl8(s) + 2Na(l) 8NaCl(s) + Fe(s)
D) 2H2O2(aq) 2H2O(l) + O2(g)
E) CO2(g) + H2(g) CO(g) + H2O(g)
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following statements relating to the glass electrode is correct?
A) The glass electrode detects hydrogen gas.
B) The glass of a glass electrode serves to conduct electrons.
C) When pH is measured, only a single electrode, the glass electrode, need be used.
D) The potential of the glass electrode varies linearly with the pH of the solution.
E) None of these choices is correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements could be prepared by electrolysis of the aqueous solution shown?
A) sodium from Na3PO4(aq)
B) sulfur from K2SO4(aq)
C) oxygen from H2SO4(aq)
D) potassium from KCl(aq)
E) nitrogen from AgNO3(aq)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Examine the following half-reactions and select the strongest reducing agent among the species listed. 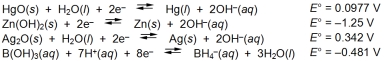
A) Hg(l)
B) Zn(s)
C) Ag(s)
D) BH4¯(aq)
E) Zn(OH) 2(s)
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following half-reactions occur in the mercury battery used in calculators. If E 1U1B1cell = 1.357 V, calculate the equilibrium constant for the cell reaction at 25 C. (Assume the stoichiometric coefficients in the cell reaction are all equal to 1.) HgO(s) + H2O(l) + 2e¯ 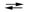 Hg(l) + 2OH¯(aq)
ZnO(s) + H2O(l) + 2e¯
Hg(l) + 2OH¯(aq)
ZnO(s) + H2O(l) + 2e¯ 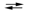 Zn(s) + 2OH¯(aq)
Zn(s) + 2OH¯(aq)
A) 9.4 * 1022
B) 7.5 *1045
C) 6.4 * 1063
D) 7.8 * 1091
E) > 9.9* 1099
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The voltaic cell made up of cobalt, copper and their M2+ ions, has E cell = 0.62 V. If E of the cathode half-cell is 0.34 V, what is E of the anode half-cell? Cu2+(aq) + Co(s) Cu(s) + Co2+(aq)
A) -0.28 V
B) -0.96V
C) 0.28 V
D) 0.96 V
E) None of these choices is correct.
G) C) and D)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
What is the value of the equilibrium constant for the cell reaction below at 25 C?
E cell = 0.30 V Sn2+(aq) + Fe(s) 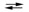 Sn(s) + Fe2+(aq)
Sn(s) + Fe2+(aq)
A) 1.2* 105
B) 1.4 * 1010
C) 8.6 * 10¯6
D) 7.1 * 10¯11
E) 2.3 * 1023
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The line notation, Al(s) | Al3+(aq) || Co2+(aq) | Co(s) , indicates that
A) Co is the reducing agent.
B) Co2+ ions are oxidized.
C) Al is the reducing agent.
D) Al3+ is the reducing agent.
E) aluminum metal is the cathode.
G) C) and E)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Predict the products of the cell reaction when a molten salt mixture of sodium bromide and calcium fluoride is electrolyzed (spectator ions are not considered to be products) .
A) calcium and bromine
B) sodium and fluorine
C) calcium bromide
D) calcium and fluorine
E) sodium and bromine
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A voltaic cell consists of an Au/Au3+ electrode (E = 1.50 V) and a Cu/Cu2+ electrode (E = 0.34 V) . Calculate [Au3+] if [Cu2+] = 1.20 M and Ecell = 1.13 V at 25 C.
A) 0.001 M
B) 0.002 M
C) 0.01 M
D) 0.02 M
E) 0.04 M
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 102
Related Exams