A) NH3 is not a nucleophile.
B) Elimination will occur.
C) NH3 is too bulky to act as a nucleophile.
D) Polyalkylation of the amine will result in multiple products.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is a typical characteristic absorption in the IR spectrum of a primary amine?
A) Two N-H absorptions at 2500-2600 cm-1
B) One N-H absorption at 2500-2600 cm-1
C) Two N-H absorptions at 3300-3500 cm-1
D) One N-H absorption at 3300-3500 cm-1
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the reagent(s) required for the following transformation. 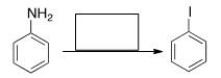
A) NaI
B) (1) NaNO2, HCl; (2) NaI
C) (1) NaNO2, HCl; (2) I2
D) I2
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the major organic product of the following reaction. 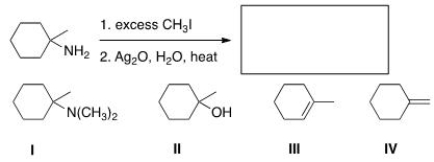
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why are alkylamines more basic than arylamines?
A) The lone pair electrons are localized in alkylamines and delocalized in arylamines.
B) The lone pair electrons are delocalized in alkylamines and localized in arylamines.
C) The lone pair electrons are less readily available in alkylamines.
D) The lone pair electrons are more readily available in arylamines.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following multistep synthesis.What is the structure of intermediate B?
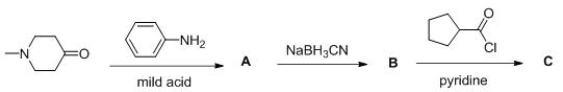
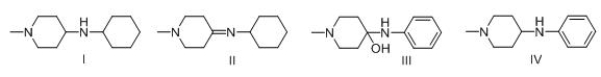
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following compounds in order of decreasing boiling point, putting the compound with the highest boiling point first. 
A) I > II > III
B) I > III > II
C) III > I > II
D) III > II > I
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name of the following compound?
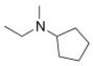
A) N-ethyl-N-methylcyclopentanamine
B) N-cyclopentyl-N-methylethanamine
C) N-methyl-N-ethylcyclopentylamine
D) N-ethyl-N-methylpentanamine
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the reagent(s) required for the following transformation. 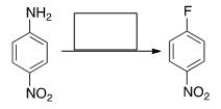
A) NaF
B) (1) NaNO2, HCl; (2) F2
C) (1) NaNO2, HCl; (2) HBF4
D) F2
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the common name of the following compound?
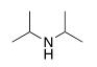
A) diisopropylamine
B) dipropylamine
C) diisopropanamine
D) dibutylamine
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name of the following compound?
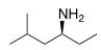
A) (S) -methyl-4-hexanamine
B) (S) -5-methyl-3-hexanamine
C) (R) -2-methyl-4-hexanamine
D) (R) -5-methyl-3-hexanamine
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the reagent(s) required for the following transformation. 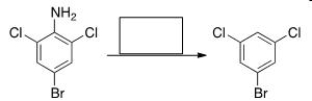
A) (1) NaNO2, HCl; (2) H2
B) (1) NaNO2, HCl; (2) H2O
C) (1) NaNO2, HCl; (2) H3PO4
D) (1) NaNO2, HCl; (2) H3PO2
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the major product of the following reaction. 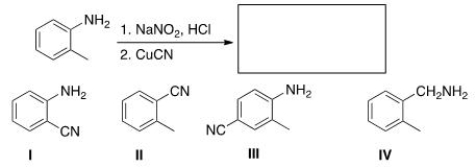
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Histamine, a vasodilator, is responsible for a wide variety of physiological effects.Rank the three nitrogen atoms in histamine in increasing order of basicity, putting least basic nitrogen atom first. 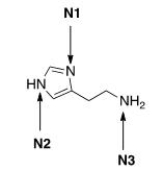
A) N2 < N1 < N3
B) N1 < N2 < N3
C) N3 < N1 < N2
D) N3 < N2 < N1
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following multistep synthesis.What is the structure of intermediate C?
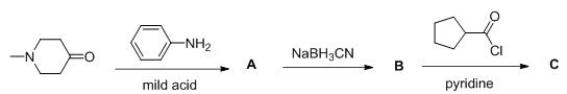
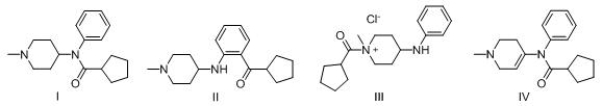
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why should the chirality of an ammonium salt with four different groups on the nitrogen atom not be ignored?
A) Because there is rapid interconversion between the two isomeric forms at room temperature.
B) Because interconversion cannot occur between the two isomeric forms at room temperature.
C) Because the compound would be a meso compound.
D) Because the compound would be a racemic mixture.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product obtained in the following reaction?
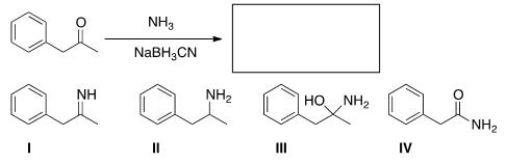
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following multistep synthesis.What is the structure of intermediate A?
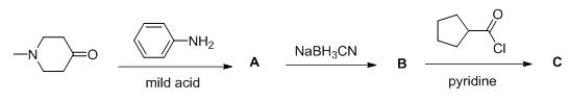
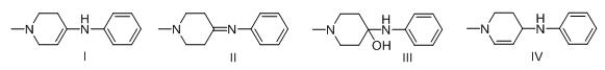
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Although an amine nitrogen atom containing an electron pair and bonded to three different groups is technically a stereogenic center, the chirality of the amine nitrogen is often ignored.Why is that?
A) Because four bonds are needed to define a stereogenic center.
B) Because chirality only exists with the tetrahedral carbon atoms.
C) Because there is usually slow interconversion between the two isomeric forms at room temperature.
D) Because there is usually rapid interconversion between the two isomeric forms at room temperature.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product obtained in the following reaction?
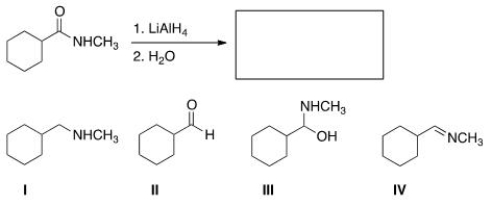
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 53
Related Exams