A) I and II
B) I and III
C) I and IV
D) II and IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following structures I-IV.Which two species represent resonance structures?
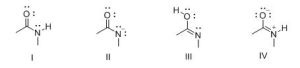
A) I and II
B) I and III
C) I and IV
D) II and IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the conjugate acid in the following reaction?

A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the conjugate acid in the following reaction?

A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following species can be both Lewis acid and Lewis base?
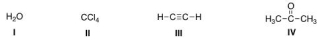
A) I, III, IV
B) I, II, IV
C) II, III, IV
D) I, II, III
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the electrophilic site in the following compounds?
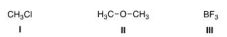
A) I = Carbon; II = carbon; III = boron.
B) I = Chlorine; II = carbon; III = boron.
C) I = Carbon; II = oxygen; III = boron.
D) I = Carbon; II = carbon; III = fluorine.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following structures I-IV.Which two species represent a conjugate acid-base pair?
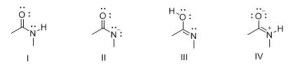
A) I and II
B) I and III
C) I and IV
D) II and III
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is the strongest acid?

A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the nucleophilic site in the following compounds?
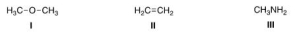
A) I = Hydrogen; II = π electrons in bond; III = nitrogen.
B) I = Oxygen; II = carbon; III = nitrogen.
C) I = Hydrogen; II = carbon; III = carbon.
D) I = Oxygen; II = π electrons in bond; III = nitrogen.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds has the lowest pKa?
A) H2O
B) H2S
C) NH3
D) CH4
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is the weakest acid?
A) HF
B) HCl
C) HBr
D) HI
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements explains why H2O is a stronger acid than CH4?
A) H2O can form hydrogen bonds while CH4 cannot.
B) H2O forms a less stable conjugate base, HO-.
C) CH4 forms a more stable conjugate base, CH3-.
D) H2O forms a more stable conjugate base, HO-.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 41 - 52 of 52
Related Exams