A) highest energy
B) largest number of possible arrangements
C) lowest number of possible arrangements
D) most symmetry
E) highest enthalpy
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is ΔS° for the following reaction? 4Cr(s) + 3O2(g) → 2Cr2O3(s) 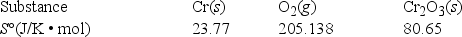
A) -549.19 J/K • mol
B) -148.26 J/K • mol
C) +148.26 J/K • mol
D) +66.22 J/K • mol
E) +871.80 J/K • mol
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In 1774 Joseph Priestly prepared oxygen by heating mercury(II) oxide according to the reaction HgO(l) <----> Hg(l) + ½O2(g) , for which ΔHo = 90.84 kJ/mol and ΔSo = 108 J/K.mol. Which of the following statements is true for this reaction?
A) The reaction is spontaneous only at low temperatures.
B) The reaction is spontaneous at all temperatures.
C) ΔGo becomes less favorable as temperature increases.
D) The reaction is spontaneous only at high temperatures.
E) The reaction is at equilibrium at 25oC and 1 atm pressure.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the process C6H6(l) <-----> C6H6(s) at a temperature above the freezing point of C6H6,
A) ΔS is positive.
B) ΔH is positive.
C) ΔG is positive.
D) ΔG = O.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
Predict the sign of ΔS for the reaction 6CO2(g) + 6H2O(g) → C6H12O6(s) + 6O2(g).
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At constant temperature and pressure, which is the correct relationship between ΔG and other thermodynamic quantities?
A) ΔG = ΔH - TΔS
B) ΔG = ΔH + TΔS
C) ΔG = ΔU - TΔS
D) ΔG = -TΔS
E) ΔG = q/T
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What term is given to the fact that the entropy of a perfect crystalline solid is zero at absolute zero?
A) 1st law of thermodynamics
B) 2nd law of thermodynamics
C) 3rd law of thermodynamics
D) crystalline lattice theory
E) absolute crystallinity
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
For a process that is spontaneous at high temperatures but not at low temperatures, the sign of ΔH is ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these species has the highest entropy (S°) at 25°C?
A) CH3OH(l)
B) CO(g)
C) MgCO3(s)
D) H2O(l)
E) Si(s)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At temperatures below 273 K, it is observed that liquid water spontaneously freezes to form solid ice. What must be true about the entropy changes of the system and surroundings for this process?
A) ΔSsys > 0, ΔSsurr > 0
B) ΔSsys < 0, ΔSsurr > 0
C) ΔSsys < 0, ΔSsurr < 0
D) ΔSsys > 0, ΔSsurr < 0
E) ΔSsys = 0, ΔSsurr > 0
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the third law of thermodynamics?
Correct Answer

verified
At 0 K all perfectly...View Answer
Show Answer
Correct Answer
verified
View Answer
Essay
State the second and third laws of thermodynamics.
Correct Answer

verified
All spontaneous processes are ...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
The complete combustion of liquid benzene is represented by the equation:
C6H6(l) + 7 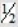 O2(g) → 6CO2(g) + 3H2O(l)
Using the data below, calculate, for this reaction
a. ΔH°
b. ΔG° at 25°C.
O2(g) → 6CO2(g) + 3H2O(l)
Using the data below, calculate, for this reaction
a. ΔH°
b. ΔG° at 25°C. 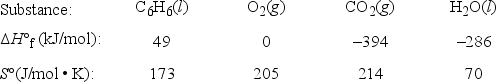
Correct Answer

verified
a. -3271 k...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
For a process where a gas condenses to a liquid, the entropy ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a chemical reaction to be spontaneous at all temperatures, which conditions must be met?
A) ΔS°rxn > 0, ΔH°rxn > 0
B) ΔS°rxn > 0, ΔH°rxn < 0
C) ΔS°rxn< 0, ΔH°rxn < 0
D) ΔS°rxn < 0, ΔH°rxn > 0
E) ΔS°rxn = 0, ΔH°rxn = 0
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is ΔS° for the following reaction? 2Cl2(g) + SO2(g) → SOCl2(g) + Cl2O(g) 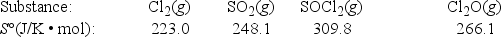
A) -118.2 J/K • mol
B) -104.8 J/K • mol
C) 104.8 J/K • mol
D) 118.2 J/K • mol
E) 1270.0 J/K • mol
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is ΔS° for the combustion of propane? C3H8(g) + 5O2(g) → 3CO2(g) + 4H2O(g) 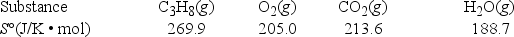
A) -100.7 J/K
B) -72.6 J/K
C) 72.6 J/K
D) 100.7 J/K
E) 877.2 J/K
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
As the molar mass of a compound increases, the entropy ________.
A) decreases
B) is constant
C) increases
E) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many types of vibrations does a bent triatomic molecule exhibit?
A) 0
B) 1
C) 2
D) 3
E) 4
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is ΔG°rxn for the following reaction? 3NO2(g) + H2O(l) → 2HNO3(l) + NO(g) 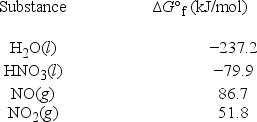
A) +8.7 kJ/mol
B) +192.2 kJ/mol
C) -178.6 kJ/mol
D) -192.2 kJ/mol
E) +639.1 kJ/mol
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 107
Related Exams