A) 21 min
B) 42 min
C) 84 min
D) 120 min
E) 101 min
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following initial rate data apply to the reaction F2(g) + 2Cl2O(g) → 2FClO2(g) + Cl2(g) . ![The following initial rate data apply to the reaction F<sub>2</sub>(g) + 2Cl<sub>2</sub>O(g) → 2FClO<sub>2</sub>(g) + Cl<sub>2</sub>(g) . Which of the following is the rate law for this reaction? A) rate = k[F<sub>2</sub>]<sup>2</sup>[Cl<sub>2</sub>O]<sup>4</sup> B) rate = k[F<sub>2</sub>]<sup>2</sup>[Cl<sub>2</sub>O] C) rate = k[F<sub>2</sub>][Cl<sub>2</sub>O] D) rate = k[F<sub>2</sub>][Cl<sub>2</sub>O]<sup>2</sup> E) rate = k[F<sub>2</sub>]<sup>2</sup>[Cl<sub>2</sub>O]<sup>2</sup>](https://d2lvgg3v3hfg70.cloudfront.net/TB8482/11eb6c5c_3a98_dea3_8d9d_750469a6f3fa_TB8482_00.jpg) Which of the following is the rate law for this reaction?
Which of the following is the rate law for this reaction?
A) rate = k[F2]2[Cl2O]4
B) rate = k[F2]2[Cl2O]
C) rate = k[F2][Cl2O]
D) rate = k[F2][Cl2O]2
E) rate = k[F2]2[Cl2O]2
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the reaction A + 2B → C, which expression is correct?
A) Δ[C]/Δt = Δ[A]/Δt
B) Δ[C]/Δt = 2Δ[B]/Δt
C) Δ[C]/Δt = -2Δ[B]/Δt
D) Δ[C]/Δt = -½Δ[B]/Δt
E) Δ[C]/Δt = -½Δ[A]/Δt
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following is an Arrhenius plot of a first-order reaction. The rate constant is measured in units of s-1. 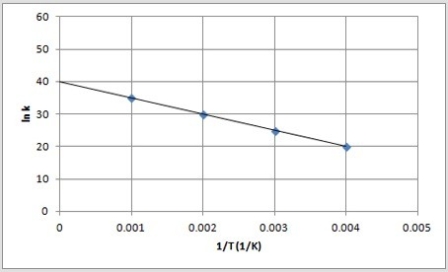 If a catalyst is added to the reaction, which could correspond to an Arrhenius plot of the catalyzed reaction?
If a catalyst is added to the reaction, which could correspond to an Arrhenius plot of the catalyzed reaction?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the correct unit for a first-order rate constant?
A) s-1
B) M • s-1
C) M • s
D) M -1 • s-1
E) M -2 • s-1
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction A + 2B → Products has been found to have the rate law, rate = k[A][B]2. While holding the concentration of A constant, the concentration of B is increased to three times its initial value. By what factor does the rate of reaction increase?
A) 3
B) 6
C) 9
D) 27
E) 30
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Below is a plot of the reaction of A decomposing to form products. 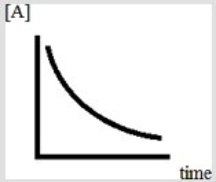 Based on the figure, which might be the order of the reaction with respect to A?
Based on the figure, which might be the order of the reaction with respect to A?
A) The reaction must only be zeroth order in A.
B) The reaction must only be first order in A.
C) The reaction must only be second order in A.
D) The reaction could be either first order in A or second order in A.
E) The reaction could be of any order in A.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
The step of the reaction mechanism which is the slowest step is called ________-________.
Correct Answer

verified
Correct Answer
verified
True/False
The rate law predicted by the following two-step mechanism is Rate = k[A][B]. A → C + B (slow) A + B → C + E (fast)
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the reaction A(g) + 2B(g) → 2C(g) + 2D(g)
The following data were collected at constant temperature. Determine the correct rate law for this reaction. ![For the reaction A(g) + 2B(g) → 2C(g) + 2D(g) The following data were collected at constant temperature. Determine the correct rate law for this reaction. A) Rate = k[A][B] B) Rate = k[A]<sup>2</sup>[B] C) Rate = k[A][B]<sup>2</sup> D) Rate = k[A] E) Rate = k[A]<sup>3</sup>](https://d2lvgg3v3hfg70.cloudfront.net/TB8482/11eb6c5c_3a96_1f80_8d9d_0fc18890c605_TB8482_00.jpg)
A) Rate = k[A][B]
B) Rate = k[A]2[B]
C) Rate = k[A][B]2
D) Rate = k[A]
E) Rate = k[A]3
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name given to the slowest step in the mechanism of a reaction?
A) Catalytic step
B) Mechanistic step
C) Rate-determining step
D) Step 1
E) Rate step
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the general reaction 5Br-(aq) + BrO3-(aq) + 6H+(aq) → 3Br2(aq) + 3H2O(aq) For this reaction, which is equal to Δ[Br2]/Δt?
A) -Δ[H2O]/Δt
B) 3Δ[BrO3-]/Δt
C) -5Δ[Br-]/Δt
D) -0.6Δ[Br-]/Δt
E) None of these choices is correct.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 121 - 132 of 132
Related Exams