A) 1
B) 2
C) 3
D) 4
E) 6
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Allene, C3H4 (shown below) , is one component of gas used for high-temperature welding. According to the valence bond model, which is a proper description of the bonding on the central carbon atom in allene? 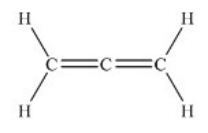
A) It shares four ![]() bonds, two with each carbon atom on either side.
bonds, two with each carbon atom on either side.
B) It shares four σ bonds, two with each carbon atom on either side.
C) It shares one σ bond and one ![]() bond with each carbon atom on either side.
bond with each carbon atom on either side.
D) It shares two σ bonds with the carbon atom on the left, and two ![]() bonds with the carbon atom on the right.
bonds with the carbon atom on the right.
E) It shares two σ bonds with the carbon atom on the right, and two ![]() bonds with the carbon atom on the left.
bonds with the carbon atom on the left.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The PCl5 molecule has
A) nonpolar bonds, and is a nonpolar molecule.
B) nonpolar bonds, but is a polar molecule.
C) polar bonds, and is a polar molecule.
D) polar bonds, but is a nonpolar molecule.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the species N2-, N2, and N2+. Which of these species will be paramagnetic?
A) N2 and N2-
B) N2+ and N2
C) N2+ and N2-
D) N2-, N2, and N2+
E) None is paramagnetic.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The BrF5 molecule has polar bonds and has a net dipole moment.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the number of lone electron pairs on the central atom of a molecule having a linear molecular geometry, such as ClF2-?
A) 1
B) 2
C) 3
D) 0
E) 4
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
How many electron domains are on the central atom for a sp2 hybridized atom?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which molecular formula corresponds to a structural formula with carbon atoms having hybridizations of sp, sp2, and sp3?
A) C3H6
B) C4H4
C) C4H6
D) C5H6
E) C5H8
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the total number of electron domains around the central atom for a molecule having a square pyramidal molecular geometry, such as ClF5?
A) 5
B) 2
C) 3
D) 4
E) 6
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to molecular orbital theory, what is the bond order of the hypofluorite ion, OF-?
A) 0
B) 0.5
C) 1
D) 1.5
E) 2
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which figure best illustrates the hybrid orbitals on carbon in benzene, C6H6?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of π bonds in phosgene, COCl2, is
A) 0.
B) 1.
C) 2.
D) 3.
E) 4.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular geometry of HOF as predicted by the VSEPR model?
A) trigonal pyramidal
B) bent
C) tetrahedral
D) linear
E) trigonal planar
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Essay
In one sentence state the basic principle of valence bond theory.
Correct Answer

verified
A covalent bond forms when the...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
What is the molecular geometry of SiF62- as predicted by the VSEPR model? 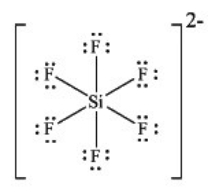
A) trigonal bipyramidal
B) octahedral
C) tetrahedral
D) seesaw
E) square pyramidal
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The hybridization of the central nitrogen atom in the molecule N2O is
A) sp.
B) sp2.
C) sp3.
D) sp3d.
E) sp3d2.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the predicted O-C-O bond angle in CO2?
A) 60°
B) 90°
C) 109.5°
D) 120°
E) 180°
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Atoms of period 3 and beyond can undergo sp3d2 hybridization, but atoms of period 2 cannot.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Indicate the type of hybrid orbitals used by the central atom in SF6.
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which figure best illustrates the hybrid orbitals on phosphorous in PH3?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 139
Related Exams