A) 4.16 V
B) -1.40 V
C) -1.32 V
D) 1.32 V
E) 1.40 V
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How long must a constant current of 50.0 A be passed through an electrolytic cell containing aqueous Cu2+ ions to produce 5.00 moles of copper metal?
A) 0.187 hours
B) 0.373 hours
C) 2.68 hours
D) 5.36 hours
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the reaction below,determine the standard free energy change,ΔG°. Cu(s) + Sn2+(aq) → Cu2+(aq) + Sn(s) E°cell = -0.48 V
A) -46.3 kJ
B) +46.3 kJ
C) -92.6 kJ
D) +92.6 kJ
F) All of the above
Correct Answer

verified
Correct Answer
verified
Essay
Explain the significance of the standard hydrogen electrode (SHE)in the tabulation of standard reduction potentials of other species.
Correct Answer

verified
The SHE is used to construct an electroc...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
What element is being reduced in the following redox reaction? MnO4⁻(aq) + H2C2O4(aq) → Mn2+(aq) + CO2(g)
A) C
B) O
C) Mn
D) H
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the criteria for a spontaneous reaction.
A) ΔG° < 0,E°cell > 0,K > 1
B) ΔG° > 0,E°cell > 0,K > 1
C) ΔG° > 0,E°cell < 0,K > 1
D) ΔG° < 0,E°cell > 0,K < 1
E) ΔG° > 0,E°cell < 0,K < 1
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What mass of silver can be plated onto an object in 33.5 minutes at 8.70 A of current? Ag⁺(aq) + e⁻ → Ag(s)
A) 19.6 g
B) 0.326 g
C) 9.78 g
D) 3.07 g
E) 0.102 g
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Match the following. -Q = K
A) Ecell = E°cell
B) Ecell = 0
C) E°cell < 0
D) Ecell < 0
E) E°cell > 0
F) Ecell > 0
H) A) and F)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The standard emf for the cell using the overall cell reaction below is +0.48 V: Zn(s) + Ni2+(aq) → Zn2+(aq) + Ni(s) The emf generated by the cell when [Ni2+] = 2.50 M and [Zn2+] = 0.100 M is ________ V.
A) 0.40
B) 0.50
C) 0.52
D) 0.56
E) 0.44
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The standard cell potential (E°) of a voltaic cell constructed using the cell reaction below is 0.76 V: Zn(s) + 2H+(aq) → Zn2+(aq) + H2(g) With PH2 = 1.0 atm and [Zn2+] = 1.0 M,the cell potential is 0.66 V.The concentration of H+ in the cathode compartment is ________ M.
A) 2.0 × 10-2
B) 4.2 × 10-4
C) 1.4 × 10-1
D) 4.9 × 101
E) 1.0 × 10-12
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine which of the following pairs of reactants will result in a spontaneous reaction at 25°C. 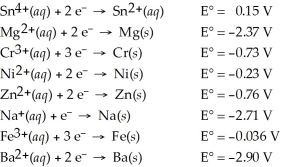
A) Sn4+(aq) + Mg(s)
B) Cr3+(aq) + Ni(s)
C) Zn(s) + Na+(aq)
D) Fe(s) + Ba2+(aq)
E) None of the above pairs will react.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many grams of chromium metal are plated out when a constant current of 8.00 A is passed through an aqueous solution containing Cr3+ ions for 320.minutes?
A) 27.6 g
B) 49.2 g
C) 82.4 g
D) 248 g
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A galvanic cell consists of a Mg2+/Mg half-cell and a standard hydrogen electrode.If the Mg2+/Mg half-cell standard cell functions as the anode,and the standard cell potential is 2.37 V,what is the standard reduction potential for the Mg2+/Mg half-cell?
A) -2.37 V
B) -1.18 V
C) +1.18 V
D) +2.37 V
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the strongest reducing agent? 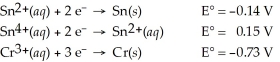
A) Sn2+(aq)
B) Cr3+(aq)
C) Sn4+(aq)
D) Cr(s)
E) Sn(s)
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the standard half-cell potentials listed below to calculate the standard cell potential for the following reaction occurring in an electrochemical cell at 25°C.(The equation is balanced. )
Sn(s) + 2 Ag⁺(aq) → Sn2+(aq) + 2 Ag(s)
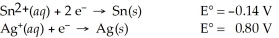
A) 1.74 V
B) 0.94 V
C) 1.08 V
D) -1.08 V
E) -1.74 V
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many electrons are transferred in the following reaction? (The reaction is unbalanced. ) Ca(s) + Cr3+(aq) → Cr(s) + Ca2+(aq)
A) 6
B) 2
C) 3
D) 1
E) 4
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What element is being reduced in the following redox reaction? H2O2(l) + ClO2(aq) → ClO2⁻(aq) + O2(g)
A) H
B) O
C) Cl
D) N
E) C
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the cell notation for the redox reaction given below. 3 Cl2(g) + 2 Fe(s) → 6 Cl⁻(aq) + 2 Fe3+(aq)
A) Cl2(g) ∣ Cl⁻(aq) ∣ Pt ∣∣ Fe(s) ∣ Fe3+(aq)
B) Cl⁻(aq) ∣ Cl2(g) ∣ Pt ∣∣ Fe3+(aq) ∣ Fe(s)
C) Fe3+(aq) ∣ Fe(s) ∣∣ Cl⁻(aq) ∣ Cl2(g) ∣ Pt
D) Fe(s) ∣ Cl2(g) ∣∣ Fe3+(aq) ∣ Cl⁻(aq) ∣ Pt
E) Fe(s) ∣ Fe3+(aq) ∣∣ Cl2(g) ∣ Cl⁻(aq) ∣ Pt
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Explain the use of a salt bridge.
Correct Answer

verified
The salt bridge allo...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
What is the reduction half-reaction for the following overall galvanic cell reaction? Zn2+(aq) + 2 Ag(s) → Zn(s) + 2 Ag+(aq)
A) Ag(s) + e- → Ag+(aq)
B) Ag+(aq) + e- → Ag(s)
C) Zn2+(aq) + 2 e- → Zn(s)
D) Zn2+(aq) + e- → Zn(s)
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 115
Related Exams