A) ( ) only
B) ( ) and *
C) ( ) only
D) ( ) and *
E) ( ) and
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
There are no -bonds in C2H6.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the number of lone pairs around the central atom and the geometry of the ion NO2-.
A) 0 lone pairs, linear
B) 1 lone pair, bent
C) 2 lone pair, bent
D) 3 lone pairs, bent
E) 3 lone pairs, linear
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A sp2 hybridized terminal oxygen atom (bonded to one other atom only) with 2 lone pairs of electrons has what type of bonding
A) 1 and 1 bonds
B) 2 and 1 bonds
C) 1 and 2 bonds
D) 0 and 2 bonds
E) 0 and 3 bonds
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of pi bonds in the molecule below is 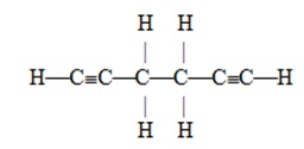
A) 2
B) 4
C) 6
D) 10
E) 15
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Indicate the type of hybrid orbitals used by the central atom in CCl4.
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The geometry of the ClF3 molecule is best described as:
A) distorted tetrahedron
B) trigonal planar
C) tetrahedral
D) T-shaped
E) trigonal pyramidal
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Shown here is the correct Lewis structure for PF5. According to VSEPR theory how many electrons are bonded to the P atom 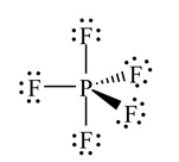
A) Four
B) Eight
C) Ten
D) Twelve
E) None of the above
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the species N2-, N2, and N2+. Which of these species will be paramagnetic
A) N2 and N2-
B) N2+ and N2
C) N2+ and N2-
D) only N2-
E) none are paramagnetic
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Indicate the type of hybrid orbitals used by the central atom in TeF4.
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The bond angles in SF5+ are expected to be
A) 90 .
B) 120 .
C) 90 and 120 .
D) 90 and 180 .
E) 90 , 120 , and 180 .
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Two py orbitals from two different atoms can interact to form a pi bonding molecular orbital.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A sp hybridized terminal nitrogen atom (bonded to one other atom only) with 1 lone pair of electrons has what type of bonding
A) 1 and 2 bonds
B) 2 and 1 bonds
C) 2 and 2 bonds
D) 0 and 2 bonds
E) 0 and 3 bonds
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The bond angles in CO32- are expected to be
A) a little less than 109.5 .
B) 109.5 .
C) a little less than 120 .
D) 120 .
E) a little more than 120 .
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The geometrical structure of PF5 and IF5 are both square pyramidal.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following correctly lists species in order of increasing bond order
A) C2 < Li2 < Be2 < N2
B) Be2 < Li2 < C2 < N2
C) N2 < Be2 < Li2 < C2
D) N2 < C2 < Li2 < Be2
E) Be2 < C2 < N2 < Li2
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Valence Bond Theory alone can be used to determine that O2 is paramagnetic.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
There are two -bonds in C2H4.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
There is one -bond in N2H2.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following molecules has a non-zero dipole moment
A) BeCl2
B) Br2
C) BF3
D) IBr
E) CO2
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 146
Related Exams