A) KI
B) KBr
C) KCl
D) KF
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A Lewis structure of boron trifluoride is shown here. How many valence electrons are shown directly around the B atom 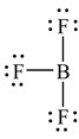
A) Eight
B) Six
C) Twenty-four
D) None
E) None of the above
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the Born-Haber cycle to calculate the lattice energy of KCl(s) given the following data: H(sublimation) K = 79.2 kJ/mol I1 (K) = 418.7 kJ/mol Bond energy (Cl-Cl) = 242.8 kJ/mol EA (Cl) = 348 kJ/mol (KCl(s) ) = -435.7 kJ/mol
A) -165 kJ/mol
B) 288 kJ/mol
C) 629 kJ/mol
D) 707 kJ/mol
E) 828 kJ/mol
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of these polar covalent bonds would have the greatest percent ionic character
A) H - Br
B) H - Cl
C) H - F
D) H - I
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ionic solids would have the largest lattice energy
A) SrO
B) NaF
C) CaBr2
D) CsI
E) BaSO4
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Define electronegativity:
A) an atoms ability to attract electrons that are shared in a chemical bond
B) an atoms ability to form an ionic bond with another atom
C) an atoms ability to donate valence electrons to another atom
D) an atoms ability to form a cation
E) an atoms ability to form double and triple bonds
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The formal charge on the bromine atom in BrO3- drawn with three single bonds is
A) -2.
B) -1.
C) 0.
D) +1.
E) +2.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a useful guideline for the application of formal charges in neutral molecules
A) A Lewis structure in which there are no formal charges is preferred.
B) Lewis structures with large formal charges are preferred.
C) The preferred Lewis structure is one in which positive formal charges are on the most electronegative atoms.
E) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The compound Al(ClO3)3 shows only ionic bonding.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the total number of lone pairs in the best Lewis structure for SOF4 that exceeds the octet rule (S is the central atom)
A) 0
B) 2
C) 14
D) 16
E) 18
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is most likely to be an ionic compound
A) CaCl2
B) CO2
C) CS2
D) SO2
E) OF2
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The Lewis structure shown here is correct for ammonia (nitrogen trihydride). 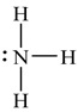
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following molecules has an atom with an expanded octet
A) HCl
B) AsCl5
C) ICl
D) NCl3
E) Cl2
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the formal charge on the singly bonded oxygens in the Lewis structure for the carbonate ion
A) -2
B) -1
C) 0
D) +1
E) +2
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Assuming the octet rule is obeyed, how many covalent bonds will a carbon atom form to give a formal charge of zero
A) 0
B) 1
C) 2
D) 3
E) 4
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the bonds below would have the greatest polarity (i.e., highest percent ionic character)
A) Si - P
B) Si - S
C) Si - Se
D) Si - Cl
E) Si - I
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the formal charge on phosphorus in a Lewis structure for the phosphate ion that satisfies the octet rule
A) -2
B) -1
C) 0
D) +1
E) +2
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The properties and chemical reactivity of a molecule is best explained by analyzing all possible resonance structures for that molecule.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Estimate the enthalpy change for the reaction 2CO + O2 2CO2 given the following bond energies. BE(C O) = 1074 kJ/mol BE(O=O) = 499 kJ/mol BE(C=O) = 802 kJ/mol
A) +2380 kJ/mol
B) +1949 kJ/mol
C) +744 kJ/mol
D) -561 kJ/mol
E) -744 kJ/mol
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A nonpolar covalent bond (i.e., pure covalent) would form in which one of the following pairs of atoms
A) Na - Cl
B) H - Cl
C) Li - Br
D) Se - Br
E) Br - Br
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 136
Related Exams