A) 4s
B) 2d
C) 3s
D) 5p
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the electron configuration for silicon?
A) 1s22s22p63s13p3
B) 1s22s22p63s23p2
C) 1s22s22p63s4
D) 1s22s22p63p4
E) 1s22s22p63s23p3
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following electron transitions would be expected to absorb any light in the Bohr model of the atom?
A) n = 1 to n = 3
B) n = 3 to n = 2
C) n = 4 to n = 2
D) n = 6 to n = 5
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the element that will lose an electron most easily, based on the periodic trend.
A) Li
B) Na
C) K
D) He
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Using the periodic table, predict the charge on the common ion of calcium.
A) +1
B) +2
C) −1
D) −2
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine which sublevel designation is legitimate.
A) 1p
B) 2p
C) 3f
D) 4z
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these choices is the electron configuration for the aluminum ion?
A) 1s22s22p63s2
B) 1s22s22p63s23p2
C) 1s22s22p63s23p1
D) 1s22s22p6
E) 1s22s22p63s23p4
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Atomic orbitals developed using quantum mechanics
A) describe regions of space in which one is most likely to find an electron.
B) describe exact paths for electron motion.
C) give a description of the atomic structure which is essentially the same as the Bohr model.
D) allow scientists to calculate an exact volume for the hydrogen atom.
E) are in conflict with the Heisenberg uncertainty principle.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
The orbital diagram for a ground-state nitrogen atom is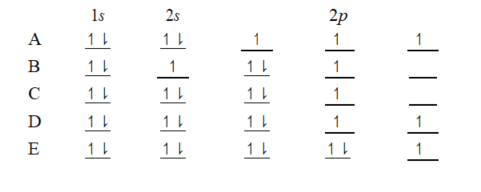
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which ground-state atom has an electron configuration described by the following orbital diagram? 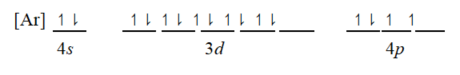
A) phosphorus
B) germanium
C) selenium
D) tellurium
E) potassium
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Not Answered
Select the element with the greatest metallic character. A)Li B)Ca C)Al D)Pb E)Cs
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The general electron configuration for noble gas atoms is
A) ns2np6.
B) ns2np5.
C) ns2np4.
D) ns2np3.
E) ns2.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Using the periodic table, predict the charge on the common ion of bromine.
A) +1
B) +2
C) −1
D) −2
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the arrangement of electromagnetic radiation which starts with the lowest energy and increases to the greatest energy.
A) radio, infrared, ultraviolet, gamma rays
B) radio, ultraviolet, infrared, gamma rays
C) gamma rays, infrared, radio, ultraviolet
D) gamma rays, ultraviolet, infrared, radio
E) infrared, ultraviolet, radio, gamma rays
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Not Answered
Select the element with the least metallic character. A)Sn B)Sr C)Tl D)Ge E)Ga
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine which sublevel designation is legitimate.
A) 1f
B) 2d
C) 3c
D) 4s
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these choices is the electron configuration for the chloride ion?
A) [Ne]3s23p4
B) [Ne]3s23p7
C) [Ar]
D) [Ar]4s1
E) [Ne]3s23p5
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many orbitals can have the 4s description in a given atom?
A) 1
B) 2
C) 3
D) 5
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
List the following types of radiation from lowest frequency to highest frequency: microwave, X ray, ultraviolet, visible, and infrared
A) microwave < infrared < visible < ultraviolet < X ray
B) X ray < ultraviolet < visible < infrared < microwave
C) visible < ultraviolet < microwave < X ray < infrared
D) infrared < X ray < microwave < ultraviolet < visible
E) infrared < visible < microwave < ultraviolet < X ray
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many electrons are there in the 3rd principal energy level (n = 3) of a phosphorus atom?
A) 3
B) 5
C) 6
D) 8
E) 10
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 107
Related Exams