A) Transition metals
B) Halogens
C) Alkali metals
D) Alkaline earth metals
E) Noble gases
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rutherford bombarded gold foil with alpha (α) particles and found that a small percentage of the particles were deflected.Which of the following was not accounted for by the model he proposed for the structure of the atom?
A) the small size of the nucleus
B) the charge on the nucleus
C) the total mass of the atom
D) the existence of protons
E) the presence of electrons outside the nucleus
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A row of the periodic table is called a(n)
A) group.
B) period.
C) isotopic mixture.
D) family.
E) subshell.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The elements in a column of the periodic table are known as
A) metalloids.
B) a period.
C) noble gases.
D) a group.
E) nonmetals.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these elements is chemically similar to magnesium?
A) Sulfur
B) Calcium
C) Iron
D) Nickel
E) Potassium
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these scientists developed the nuclear model of the atom?
A) John Dalton
B) Robert Millikan
C) J.J.Thomson
D) Henry Moseley
E) Ernest Rutherford
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a nonmetal?
A) Lithium, Li, Z = 3
B) Bromine, Br, Z = 35
C) Mercury, Hg, Z = 80
D) Bismuth, Bi, Z = 83
E) Sodium, Na, Z = 11
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Bromine is the only nonmetal that is a liquid at room temperature.Consider the isotope bromine-81, 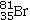 .Select the combination which lists the correct atomic number, number of neutrons, and mass number, respectively.
.Select the combination which lists the correct atomic number, number of neutrons, and mass number, respectively.
A) 35, 46, 81
B) 35, 81, 46
C) 81, 46, 35
D) 46, 81, 35
E) 35, 81, 116
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a metal?
A) Nitrogen, N, Z = 7
B) Phosphorus, P, Z = 15
C) Arsenic, As, Z = 33
D) Thallium, Tl, Z = 81
E) Silicon, Si, Z = 14
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
C(graphite) and C(diamond) are examples of:
A) isotopes of carbon.
B) allotropes of carbon.
C) the law of definite proportions.
D) different carbon ions.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Lithium forms compounds which are used in dry cells, storage batteries, and in high-temperature lubricants.It has two naturally occurring isotopes, 6Li (isotopic mass = 6.015123 amu) and 7Li (isotopic mass = 7.016005 amu) .Lithium has an atomic mass of 6.9412 amu.What is the percent abundance of lithium-6?
A) 92.53%
B) 86.65%
C) 49.47%
D) 7.47%
E) 6.015%
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What elements and groups have properties that are most similar to those of chlorine?
A) F, Br, I, and nonmetals in Group 7A
B) Cl, K, C, and metals in Group 1B
C) N, P, As, and lanthanides
D) He, Ne, Xe, and nonmetals in Group 7A
E) O, S, and P
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the term used for findings that are summarized based on a pattern or trend?
A) Law
B) Hypothesis
C) Theory
D) Phenomena
E) Prediction
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which element would be expected to have properties similar to calcium?
A) Ba
B) K
C) Sc
D) Na
E) Rb
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What element is represented by X in the atomic symbol notation ? 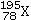
A) Iridium
B) Platinum
C) Palladium
D) Selenium
E) Magnesium
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Two isotopes of an element differ only in their
A) symbol.
B) atomic number.
C) atomic mass.
D) number of protons.
E) number of electrons.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the number of protons, electrons, and neutrons for the isotope gold-118.The symbol for gold is Au.
A) 118 protons, 118 electrons, 79 neutrons
B) 79 protons, 79 electrons, 118 neutrons
C) 79 protons, 79 electrons, 39 neutrons
D) 118 protons, 118 electrons, 39 neutrons
E) 79 protons, 39 electrons, 118 neutrons
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
When applying the scientific method, it is important to avoid any form of hypothesis.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following statements about atoms and subatomic particles is correct?
A) Rutherford discovered the atomic nucleus by bombarding gold foil with electrons
B) The proton and the neutron have identical masses.
C) The neutron's mass is equal to that of a proton plus an electron.
D) A neutral atom contains equal numbers of protons and electrons.
E) An atomic nucleus contains equal numbers of protons and neutrons.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these elements exhibits chemical behavior similar to that of potassium?
A) magnesium
B) sodium
C) sulfur
D) chlorine
E) iron
G) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 50
Related Exams