A) 7
B) 3
C) 4n
D) ![]()
E) 15p
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What would the atom ratio of 206Pb to 238U be in a uranium mineral from a rock that is 1.0 × 109 years old? t1/2(238U) = 4.5 × 109 yr.
A) 0.14
B) 0.16
C) 0.22
D) 0.86
E) 1.16
G) C) and D)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Consider the following decay series: 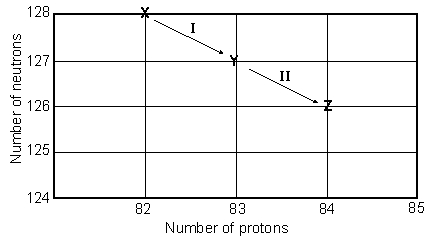 What type of nuclear process occurs at the transformation labeled II?
What type of nuclear process occurs at the transformation labeled II?
A) ( emission)
B) ( emission)
C) positron emission
D) electron capture
E) gamma radiation
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The energy released by the sun is the result of
A) natural radioactivity.
B) nuclear fusion.
C) combustion of hydrogen.
D) photosynthesis.
E) nuclear fission.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the following reaction, identify X. 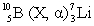
A) ( )
B) n
C) p
D) (![]() )
)
E) ( )
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The energy equivalent of a mass defect of - 0.1620 amu is which of the following?
A) - 1.458 x 1016 J
B) - 2.421 x 10-8 J
C) - 2.421 x 10-11 J
D) - 2.421 x 1013 J
E) - 1.458 x 10-11 J
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
The alpha particles emitted by radon-222 have an energy of 8.8 × 10-13 J.If a 200.g Pb brick absorbs 1.0 × 1010 alpha particles from radon decay, what dose in rads will the brick absorb?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What fraction of radioactive atoms remains in a sample after six half-lives?
A) zero
B) 1/6
C) 1/16
D) 1/32
E) 1/64
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following decay series: 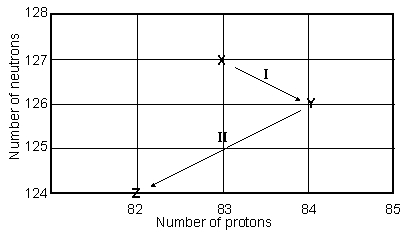 What type of nuclear process occurs at the transformation labeled II?
What type of nuclear process occurs at the transformation labeled II?
A) ( emission)
B) ( emission)
C) positron emission
D) electron capture
E) gamma radiation
G) C) and E)
Correct Answer

verified
A
Correct Answer
verified
Short Answer
What type of nuclear process is illustrated below? 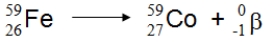
Correct Answer

verified
beta decay...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Consider the following decay series: 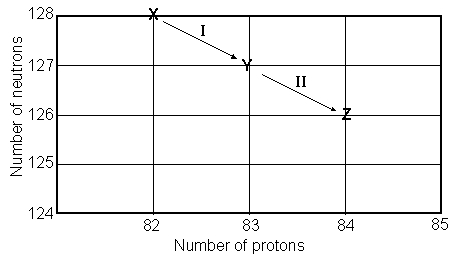 What is the complete element symbol for the product labeled Y?
What is the complete element symbol for the product labeled Y?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
The isotope of iodine with mass number 128 undergoes beta decay with a half-life of 25.00 minutes.How long will it take for 93.75% of a sample of pure 128 I to decay?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A typical radius of an atomic nucleus is about
A) 100 µm
B) 5000 mm
C) 100 nm
D) 5 × 10-3 pm
E) 500 pm
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The heaviest known isotope of hydrogen is called tritium, 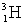 .It decays by beta emission, and has a half-life of 12.3 years.What fraction of a tritium sample will remain after 5.20 years?
.It decays by beta emission, and has a half-life of 12.3 years.What fraction of a tritium sample will remain after 5.20 years?
A) 0.0210
B) 0.746
C) 3.41
D) 0.254
E) 0.423
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A balanced nuclear equation representing the beta emission of iodine-131 is which of the following?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) None of the above
Correct Answer

verified
Correct Answer
verified
Essay
Balance the equation 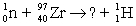 .
.
Correct Answer

verified
11ea7cbf_8f44_a6ac_a2ab_f9c90e28d3dd_TB3246_00
Correct Answer
verified
Multiple Choice
What fraction of radioactive atoms remains in a sample after five half-lives?
A) zero
B) 1/6
C) 1/16
D) 1/32
E) 1/64
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The only stable isotope of aluminum is aluminum-27.What type of radioactive decay should be expected from 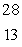 Al?
Al?
A) ![]() H
H
B) ![]() n
n
C) ![]()
D) ![]()
E) ![]() He
He
G) All of the above
Correct Answer

verified
Correct Answer
verified
Essay
Complete and balance the nuclear equation 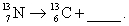
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Find the nuclear binding energy of potassium-40 (atomic mass = 39.9632591 amu) in units of joules per nucleon.[Data: neutron mass = 1.674928 × 10-24 g; proton mass = 1.672623 × 10-24g; electron mass = 9.109387 × 10-28 g; NA = 6.0221367 × 1023 /mol; c = 2.99792458 × 108 m/s]
A) 1.37 × 10-12 J/nucleon
B) 5.48 × 10-11 J/nucleon
C) 5.64 × 10-11 J/nucleon
D) 1.41 × 10-12 J/nucleon
E) 2.97 × 10-12 J/nucleon
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 133
Related Exams