A) linear
B) trigonal planar
C) bent
D) tetrahedral
E) trigonal pyramidal
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of pi bonds in the molecule below is 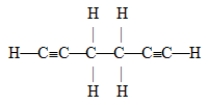
A) 2
B) 4
C) 6
D) 10
E) 15
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following molecules is polar?
A) PBr5
B) CCl4
C) BrF5
D) XeF2
E) XeF4
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Ibuprofen is used as an analgesic for the relief of pain, and also to help reduce fever. 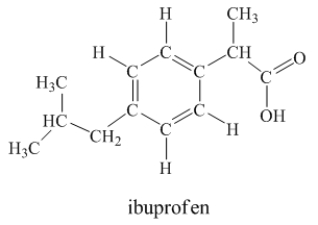 How many sigma bonds and pi bonds are contained in a ibuprofen molecule?
How many sigma bonds and pi bonds are contained in a ibuprofen molecule?
Correct Answer

verified
33 sigma b...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
Indicate the number of -bonds in N2H2.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Indicate the type of hybrid orbitals used by the central atom in SF6.
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The BrF5 molecule has polar bonds and has a net dipole moment.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The bond angles in CO32- are expected to be
A) a little less than 109.5°.
B) 109.5°.
C) a little less than 120°.
D) 120°.
E) a little more than 120°.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
N,N-diethyl-m-tolumide (DEET) is the active ingredient in many mosquito repellents.What is the hybridization state of the nitrogen atom in the structure of DEET shown below? 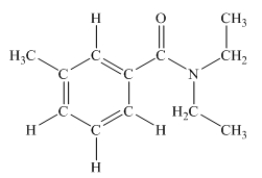
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The geometry of the CS2 molecule is best described as
A) linear.
B) trigonal planar.
C) tetrahedral.
D) bent.
E) trigonal pyramidal.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Use VSEPR theory to explain why the water molecule is bent, rather than linear.
Correct Answer

verified
About the central oxygen atom are two lo...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
If a triatomic molecule is linear, then the hybridization of the central atom will be
A) sp
B) sp2
C) sp or sp3
D) sp or sp3d
E) sp2 or sp3d2
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following species has the largest dipole moment (i.e., is the most polar) ?
A) CH4
B) CH3Br
C) CH3Cl
D) CH3F
E) CH3I
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the hybridization on the central atom in NO3-?
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the geometry and polarity of the CS2 molecule.
A) linear, polar
B) linear, nonpolar
C) tetrahedral, nonpolar
D) bent, nonpolar
E) bent, polar
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the hybridization of the As atom in the AsF5 molecule?
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Use VSEPR theory to predict the molecular geometry of SF4 (sulfur tetrafluoride).
Correct Answer

verified
distorted ...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Indicate the type of hybrid orbitals used by the central atom in TeF4.
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the molecular geometry and polarity of the SO2 molecule.
A) linear, polar
B) linear, nonpolar
C) bent, polar
D) bent, nonpolar
E) None of the above.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
N,N-diethyl-m-tolumide (DEET)is the active ingredient in many mosquito repellents. 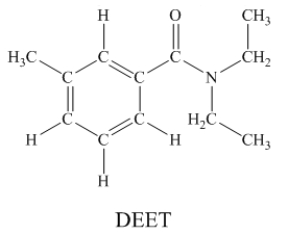 How many sigma bonds and pi bonds are contained in a DEET molecule?
How many sigma bonds and pi bonds are contained in a DEET molecule?
Correct Answer

verified
31 sigma b...View Answer
Show Answer
Correct Answer
verified
View Answer
Showing 101 - 120 of 122
Related Exams