A) Al and Br2
B) Al and O2
C) H2 and O2
D) H2 and Br2
E) Al and H2
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How long will it take to produce 78 g of Al metal by the reduction of Al3+ in an electrolytic cell with a current of 2.0 A?
A) 0.01 s
B) 420 s
C) 13 h
D) 116 h
E) 1.0 1012 s
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Calculate the cell emf for the following reaction at 25°C: 2Ag+(0.010 M)+ H2(1 atm) 2Ag(s)+ 2H+(pH = 6.0)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider an electrochemical cell constructed from the following half cells, linked by an external circuit and by a KCl salt bridge. • an Al(s) electrode in 1.0 M Al(NO3) 3 solution • a Pb(s) electrode in 1.0 M Pb(NO3) 2 solution The balanced overall (net) cell reaction is
A) Pb(s) + Al3+(aq) Pb2+(aq) + Al(s.)
B) 3Pb(s) + 2Al3+(aq) 3Pb2+(aq) + 2Al(s) .
C) 3Pb2+(aq) + 2Al(s) 3Pb(s) + 2Al3+(aq) .
D) Pb2+(aq) + Al(s) Pb(s) + Al3+(aq) .
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Given the following standard reduction potentials, 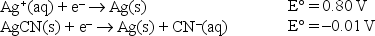 calculate the solubility product of AgCN at 25°C.
calculate the solubility product of AgCN at 25°C.
A) 4.3 10-14
B) 2.3 1013
C) 2.1 10-14
D) 5.1 1013
E) None of these
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following reagents is capable of transforming Fe3+ (1 M) to Fe2+ (1 M) ?
A) H2(1 atm)
B) NO3- (1 M)
C) O2(1 atm)
D) Br- (1 M)
E) H+ (1 M)
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If the measured voltage of the cell Zn(s) | Zn2+(aq) || Ag+(aq) | Ag(s) is 1.37 V when the concentration of Zn2+ ion is 0.010 M, what is the Ag+ ion concentration?
A) 2.5 M
B) 4.0 10-9 M
C) 6.2 10-3 M
D) 2.6 10-51 M
E) 6.2 10-5 M
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Consider the reaction Fe + Sn2+(1 10-3 M) Fe2+(1.0 M)+ Sn. Calculate the voltage theoretically generated by the cell.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The half-reaction that occurs at the cathode during electrolysis of an aqueous CuCl2 solution is
A) Cu+ + e- Cu.
B) Cu2+ + e- Cu+.
C) 2H2O + 2e- H2 + 2OH-.
D) Cl2 + 2e- 2Cl-.
E) 2Cl- Cl2 + 2e-.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the reaction Ni2+(aq) + 2Fe2+(aq) Ni(s) + 2Fe3+(aq) , the standard cell potential E°cell is
A) +2.81 V.
B) +1.02 V.
C) +0.52 V.
D) -1.02 V.
E) -2.81 V.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A certain electrochemical cell has for its cell reaction: Zn + HgO ZnO + Hg Which is the half-reaction occurring at the anode?
A) HgO + 2e- Hg + O2-
B) Zn2+ + 2e- Zn
C) Zn Zn2+ + 2e-
D) ZnO + 2e- Zn
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these metals will not be oxidized in hydrochloric acid under standard conditions at 25°C?
A) Al
B) Fe
C) Ag
D) Ni
E) Mg
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following reagents is capable of transforming Cu(s) to Cu2+ (1 M) ?
A) I- (1 M)
B) Ni(s)
C) Ag+ (1 M)
D) Al3+ (1 M)
E) H+ (1 M)
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Complete and balance the following redox equation. When properly balanced using the smallest whole-number coefficients, the coefficient of S is H2S + HNO3 S + NO (acidic solution)
A) 1
B) 2
C) 3
D) 5
E) 6
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the cell emf for the following reaction at 25°C: 2Ag+(0.010 M) + H2(1 atm) 2Ag(s) + 2H+(pH = 10.0)
A) 1.04 V
B) 1.27 V
C) 0.92 V
D) 0.56 V
E) 0.80 V
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider an electrochemical cell with the following cell reaction where all reactants and products are at standard-state conditions: Cu2+(aq) + H2(g) Cu(s) + 2H+(aq) .Predict the effect on the emf of this cell of adding NaOH solution to the hydrogen half-cell until the pH equals 7.0.
A) The emf will increase.
B) The emf will decrease.
C) No change in the emf will be observed.
E) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following standard reduction potentials in acid solution: 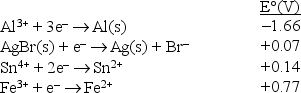 The strongest reducing agent among those shown above is
The strongest reducing agent among those shown above is
A) Fe3+.
B) Fe2+.
C) Br-.
D) Al3+.
E) Al.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the electrochemical cell Pt(s) | H2(1 atm) | H+(1 M) || Cu2+(1 M) | Cu(s) , which one of the following changes will cause an increase in the cell voltage?
A) Lower the H2(g) pressure.
B) Increase the size/mass of the copper electrode.
C) Lower the H+(aq) concentration.
D) Decrease the concentration of Cu2+ ion.
E) None of the above.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider an electrochemical cell based on the spontaneous reaction 2AgCl(s) + Zn(s) 2Ag(s) + 2Cl- + Zn2+. If the zinc ion concentration is kept constant at 1 M, and the chlorine ion concentration is decreased from 1 M to 0.001 M, the cell voltage should
A) increase by 0.06 V.
B) increase by 0.18 V.
C) decrease by 0.06 V.
D) decrease by 0.18 V.
E) increase by 0.35 V.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A current of 0.80 A was applied to an electrolytic cell containing molten CdCl2 for 2.5 hours.Calculate the mass of cadmium metal deposited.
A) 3.2 10-7 g
B) 1.2 10-3 g
C) 4.2 g
D) 8.4 g
E) 16.8 g
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 138
Related Exams