A) l = 3,m = 0
B) l = 3,m = 1
C) l = 3,m = 2
D) l = 3,m = 3
E) l = 3,m = 4
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A photon of wavelength 1.17 m is emitted by a He+ when the electron jumps from the n = 7 state.What is the final state of the electron?
A) 1
B) 2
C) 3
D) 4
E) 5
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Bohr model of hydrogen is based on the quantization of
A) electrical charge.
B) orbital angular momentum.
C) electric force.
D) magnetic force.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the energy of the photon required to knock the electron out of a singly ionized Helium atom where the electron was in the n = 4 state?
A) 3.4 eV
B) 1.7 eV
C) 10.2 eV
D) 51 eV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A hydrogen atom is in a quantum state with principal quantum number n = 3.The atom emits a photon with a wavelength of 660 nm.Determine the maximum possible orbital angular momentum of the electron after emission.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For an electron in the ground state of a hydrogen atom,what is the probability of finding that electron within a Bohr radius of the proton? The ground state wave function for hydrogen is 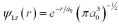 .
.
A) 0.124
B) 0.261
C) 0.323
D) 0.626
E) 0.752
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
About how many photons are emitted per second by a yellow (600 nm) 60-mW laser?
A) 2 * 1015
B) 3 * 1019
C) 3 * 1018
D) 2 * 1012
E) 2 * 1017
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the energy of the orbiting electron in a hydrogen atom with a quantum number of 45?
A) +3.8 * 10-3 eV
B) -4.2 * 10-3 eV
C) -6.7 * 10-3 eV
D) +4.5 * 10-3 eV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The wavelength of the  line of the Lyman group for the hydrogen spectrum is
line of the Lyman group for the hydrogen spectrum is
A) 93 nm.
B) 106 nm.
C) 250 nm.
D) 397 nm.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the angular momentum of an electron in an l = 4 level of the correct quantum mechanical hydrogen atom?
A) 1/2 Js
B) 0.5 * 105 Js
C) 0.1 * 1025 Js
D) 6.6 * 10-23 Js
E) 4.6 * 10-34 Js
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A laser (with wavelength of  ) excites the electron in a hydrogen atom from its ground state to its first excited state.What laser wavelength is required to raise the electron from the first excited state to its next lowest excited state?
) excites the electron in a hydrogen atom from its ground state to its first excited state.What laser wavelength is required to raise the electron from the first excited state to its next lowest excited state?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A UV laser emits light that has a wavelength 350 nm.If 1.23 * 1018 photons are emitted in 10 ns,what is the power of the laser?
A) 77 MW
B) 92 MW
C) 83 MW
D) 70 MW
E) 5.0 MW
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the Bohr hydrogen atom,how many antinodes are there in the electron standing wave with a quantum number of 6?
A) 12
B) 6
C) 3
D) 5
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
He+ is a helium atom with one electron missing.Treating this like a hydrogen atom,what is the first excited state of this atom?
A) -6.40 eV
B) -13.6 eV
C) -54.4 eV
D) -76.8 eV
E) -152.2 eV
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A group of hydrogen-like atoms (atoms with only one electron) are in their ground state.Light of increasing intensity is shone at these atoms until the light becomes absorbed by the atoms.The lowest frequency of light when this occurs is 2.22 * 1016 Hz.What is the ground state energy of these atoms?
A) 91.8 eV
B) 13.6 eV
C) 218 eV
D) 122 eV
E) 54 eV
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Find the frequency of light emitted by a hydrogen atom when its electron makes the transition from the n = 5 state to the n = 2 state.
A) 2.3 * 106 Hz
B) 1.1 * 107 Hz
C) 6.9 * 1014 Hz
D) 3.3 * 1015 Hz
E) 3.8 * 1015 Hz
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the shortest wavelength photon that can be emitted by singly ionized helium (He+) ?
A) 23 nm
B) 46 nm
C) 54 nm
D) 91 nm
E) 365 nm
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Pfund series results from emission/absorption of photons due to transitions of electrons in a hydrogen to/from the n=5 energy level from/to higher energy levels.What is the shortest wavelength photon emitted in the Pfund series?
A) 658 nm
B) 2.29 µm
C) 2.74 µm
D) 3.68 µm
E) 12.4 µm
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In a "muonic" atom an electron is replaced by a muon of mass 105.66 MeV which orbits a proton.What is the ground-state energy of the muon in this type of atom?
A) -281 eV
B) -632 eV
C) -1264 eV
D) -2528 eV
E) -3254 eV
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The electron in a singly ionized helium atom,which consists of two protons,two neutrons and only one electron,makes a transition from the n = 2 state to the n = 1 state,emitting a single photon in the process.What is the energy of this photon?
A) 3.40 eV
B) 10.2 eV
C) 13.6 eV
D) 40.8 eV
E) None are correct.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 72
Related Exams