A) H bonds must exist between solvent and solute.
B) strong ion-dipole forces must exist in the solution.
C) the solute must be a gas.
D) the entropy of the solution is immaterial.
E) the entropy of the solution must be greater than that of its pure components.
G) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
In general,water is a good solvent for both polar and non-polar compounds.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Heats of solution may be either positive or negative.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Henry's Law constant (k) for carbon monoxide in water at 25°C is 9.71 10-4 mol/(L·atm) .How many grams of CO will dissolve in 1.00 L of water if the partial pressure of CO is 2.75 atm?
A) 3.53 10-4 g
B) 2.67 10-3 g
C) 9.89 10-3 g
D) 7.48 10-2 g
E) None of these choices is correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Barbiturates are synthetic drugs used as sedatives and hypnotics.Barbital ( 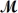 = 184.2 g/mol) is one of the simplest of these drugs.What is the boiling point of a solution prepared by dissolving 42.5 g of barbital in 825 g of acetic acid?
Kb = 3.07°C/m,boiling point of pure acetic acid = 117.9°C
= 184.2 g/mol) is one of the simplest of these drugs.What is the boiling point of a solution prepared by dissolving 42.5 g of barbital in 825 g of acetic acid?
Kb = 3.07°C/m,boiling point of pure acetic acid = 117.9°C
A) 117.0°C
B) 117.7°C
C) 118.1°C
D) 118.8°C
E) >120°C
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hexachlorophene is used as a disinfectant in germicidal soaps.What mass of hexachlorophene ( 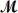 = 406.9 g/mol) must be added to 125 g of chloroform to give a solution with a boiling point of 62.60°C?
Kb = 3.63°C/m,boiling point of pure chloroform = 61.70°C
= 406.9 g/mol) must be added to 125 g of chloroform to give a solution with a boiling point of 62.60°C?
Kb = 3.63°C/m,boiling point of pure chloroform = 61.70°C
A) 12.6 g
B) 17.2 g
C) 31.0 g
D) 34.4 g
E) 101 g
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When two pure substances are mixed to form a solution
A) heat is released.
B) heat is absorbed.
C) there is an increase in entropy.
D) there is a decrease in entropy.
E) entropy is conserved.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What volume of concentrated (14.7 M) phosphoric acid is needed to prepare 25.0 L of 3.0 M H3PO4?
A) 0.20 L
B) 0.57 L
C) 1.8 L
D) 3.6 L
E) 5.1 L
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the freezing point of a solution which contains 0.31 mol of sucrose in 175 g of water.Kf = 1.86°C/m
A) 3.3°C
B) 1.1°C
C) 0.0°C
D) -1.1°C
E) -3.3°C
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A solution of sucrose (sugar) in water is in equilibrium with solid sucrose.If more solid sucrose is now added,with stirring,
A) the concentration of the solution will increase.
B) the concentration of the solution will decrease.
C) the concentration of the solution will remain the same.
D) the volume of solution will increase.
E) a supersaturated solution will be produced.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following aqueous solutions should demonstrate the most non-ideal behavior?
A) 0.1 M NaI
B) 0.2 M CuSO4
C) 0.5 M KBr
D) 2.0 M CuCl2
E) 2.5 M CsCl
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The concentration of iodine in sea water is 60.parts per billion by mass.If one assumes that the iodine exists in the form of iodide anions,what is the molarity of iodide in sea water? (The density of sea water is 1.025 g/mL. )
A) 4.8 10-13 M
B) 4.8 10-10 M
C) 4.8 10-7 M
D) 4.7 10-4 M
E) 4.7 10-1 M
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
A 0.137 m solution of sodium ferrocyanide (NaFe(CN)6.10H2O)in water has a freezing point depression of 0.755°C.Calculate the van't Hoff factor (i)under these conditions.(Kf = 1.86°C/m)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 73 of 73
Related Exams