A) combination
B) decomposition
C) displacement
D) acid-base
E) none of the above
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the product(s) for the following reaction. BaO(s) + CO2(g)
A) Ba(s) + CO3(g)
B) BaCO3(s)
C) BaO(s) + C(s)
D) Ba(s) + CO32-(s)
E) No reaction occurs.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Essay
Aluminum metal dissolved in hydrochloric acid as follows: 2Al(s) + 6HCl(aq) 2AlCl3(aq) + 3H2(g) a. What is the minimum volume of 6.0 M HCl(aq) needed to completely dissolve 3.20 g of aluminum in this reaction? b. What mass of AlCl3 would be produced by complete reaction of 3.20 g of aluminum?
Correct Answer

verified
a. 59.3 mL...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Sodium chlorate is used as an oxidizer in the manufacture of dyes, explosives, and matches. Calculate the mass of solute needed to prepare 1.575 L of 0.00250 M NaClO3 ( 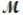 = 106.45 g/mol) .
= 106.45 g/mol) .
A) 419 g
B) 169 g
C) 0.419 g
D) 0.169 g
E) 0.00394 g
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Essay
Write down the oxidation number of the indicated atom in each of the following formulas: a. Si in SiO2 b. Cl in ClO2- c. Mn in KMnO4 d. C in C6H12O6
Correct Answer

verified
a. +4
b. +...View Answer
Show Answer
Correct Answer
verified
b. +...
View Answer
Multiple Choice
Select the classification for the following reaction. Fe2+(aq) + 2OH-(aq) Fe(OH) 2(s)
A) precipitation
B) acid-base
C) redox
D) decomposition
E) none of the above
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many moles of H+(aq) ions are present in 1.25 L of 0.75 M nitric acid?
A) 0.60 mol
B) 0.75 mol
C) 0.94 mol
D) 1.7 mol
E) 1.9 mol
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a strong acid?
A) H3PO4
B) HNO3
C) HF
D) CH3COOH
E) H2O
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the statements below correctly describes the combustion of glucose, shown below? C6H12O6 + 6O2 6CO2 + 6H2O
A) Hydrogen in C6H12O6 is being reduced.
B) Oxygen in O2 is being oxidized.
C) Hydrogen in C6H12O6 is the reducing agent.
D) Oxygen in C6H12O6 is the oxidizing agent.
E) Carbon in C6H12O6 is being oxidized.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the best statement relating to the following reaction: 2MnO2(s) + KClO3(aq) + 2KOH(aq) 2KMnO4(aq) + KCl(aq) + H2O(l)
A) Mn in MnO2 is oxidized.
B) O in KClO3 is the oxidizing agent.
C) K in KClO3 is the reducing agent.
D) H in KOH is oxidized.
E) Cl in KClO3 is the reducing agent.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
All acid-base reactions produce a salt and water as the only products.
B) False
Correct Answer

verified
Correct Answer
verified
Essay
Complete and balance the equation for the following acid-base reaction. Ca(OH)2(aq) + HCl(aq)
Correct Answer

verified
Ca(OH)2(aq) + 2HCl(aq...View Answer
Show Answer
Correct Answer
verified
View Answer
True/False
A particular reaction may be both a precipitation and an acid-base (neutralization) reaction.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is not a redox reaction?
A) 2Na(s) + 2H2O(l) 2NaOH(aq) + H2(g)
B) H2(g) + Cl2(g) 2HCl(g)
C) 2H2O2(aq) 2H2O(l) + O2(g)
D) Fe2O3(s) + 3H2SO4(aq) Fe2(SO4) 3(aq) + 3H2O(l)
E) 2KMnO4(aq) + 10FeSO4(aq) + 8H2SO4(aq) K2SO4(aq) + 2MnSO4(aq) + 5Fe2(SO4) 3(aq) + 8H2O(l)
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is not a redox reaction?
A) 2H2O2(aq) 2H2O(l) + O2(g)
B) N2(g) + 3H2(g) 2NH3(g)
C) BaCl2(aq) + K2CrO4(aq) BaCrO4(aq) + 2KCl(aq)
D) 2Al(s) + Fe2O3(s) Al2O3(s) + 2Fe(s)
E) 2H2O(g) 2H2(g) + O2(g)
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following solutions will be the best conductor of electrical current?
A) methyl alcohol, CH3OH(aq)
B) glucose, C6H12O6(aq)
C) potassium chloride, KCl(aq)
D) bromine, Br2(aq)
E) ethylene glycol, C2H6O2(aq)
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the products by completing a balanced equation for the following decomposition reaction. CaCl2(l) 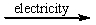 ?
?
A) CaCl2(l) ![]() Ca(l) + 2Cl-(l)
Ca(l) + 2Cl-(l)
B) CaCl2(l) ![]() Ca2+(l) + Cl2(g)
Ca2+(l) + Cl2(g)
C) CaCl2(l) ![]() Ca2+(l) + 2Cl-(l)
Ca2+(l) + 2Cl-(l)
D) CaCl2(l) ![]() Ca(l) + Cl2(g)
Ca(l) + Cl2(g)
E) CaCl2(l) ![]() CaCl(l) + Cl-(l)
CaCl(l) + Cl-(l)
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the classification for the following reaction. NH3(aq) + HNO3(aq) NH4NO3(aq)
A) precipitation
B) acid-base
C) redox
D) decomposition
E) none of the above
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
The combustion of an element is always a combination reaction.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Magnesium carbonate, MgCO3, is insoluble. Identify the spectator ions when aqueous solutions of sodium carbonate and magnesium chloride are combined.
A) Mg2+ and CO32-
B) Na+ and Cl-
C) Mg2+ and Cl-
D) Na+ and CO32-
E) None of the above correctly identifies the spectator ions.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 124
Related Exams