A) III < II < IV < I
B) III < IV < II < I
C) I < IV < II < III
D) I < II < III < IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the correct IUPAC name of the following compound? 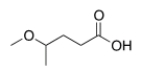
A) 2-Methoxypentanoic acid
B) 2-Methoxybutanoic acid
C) 4-Methoxybutanoic acid
D) 4-Methoxypentanoic acid
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What two groups make up the carboxylic acid group (RCOOH) ?
A) Carbon dioxide and hydrogen
B) Carbonyl and hydroxyl
C) Carbon monoxide and hydroxyl
D) Carbonyl oxide and hydrogen
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of decreasing acidity, putting the most acidic first. 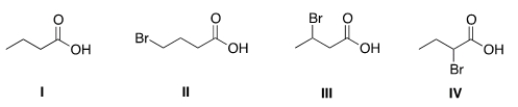
A) IV > III > II > I
B) IV > I > III > II
C) I > II > III > IV
D) IV > II > III > I
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the correct IUPAC name of the following compound? 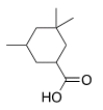
A) 3,5,5-Trimethylcyclohexanecarboxylic acid
B) 3,3,5-Trimethylcyclohexanecarboxylic acid
C) 3,3,5-Trimethylcyclohexanoic acid
D) 3,5,5-Trimethylcyclohexanoic acid
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of decreasing acidity, putting the most acidic first. 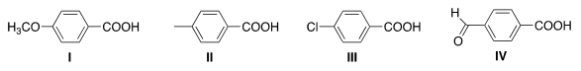
A) III > IV > I > II
B) IV > III > I > II
C) I > II > III > IV
D) IV > III > II > I
F) A) and B)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
What would happen if a mixture of benzoic acid and cyclohexanol dissolved in CH2Cl2 is treated with aqueous NaOH solution?
A) Benzoic acid would remain in the CH2Cl2 layer, and cyclohexanol would dissolve in the aqueous layer.
B) Benzoic acid would dissolve in the aqueous layer while cyclohexanol would remain in the CH2Cl2 layer.
C) The salt of benzoic acid would dissolve in the aqueous layer while cyclohexanol would remain in the CH2Cl2 layer.
D) The salt of benzoic acid would remain in the CH2Cl2 layer while cyclohexanol would dissolve in the aqueous layer.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the correct IUPAC name of the following compound? 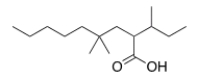
A) 2-sec-Butyl-4,4-dimethylnonanoic acid
B) 4,4-Dimethyl-2-isobutylnonanoic acid
C) 4,4-Dimethyl-2-sec-butylnonanoic acid
D) 2-Isobutyl-4,4-dimethylnonanoic acid
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What would happen if a mixture of benzoic acid (C6H5COOH) and NaCl is added to a separatory funnel containing H2O and CH2Cl2?
A) The benzoic acid would dissolve in the water layer and the NaCl would dissolve in the organic layer.
B) The benzoic acid would dissolve in the organic layer and the NaCl would dissolve in the water layer.
C) Both benzoic acid and NaCl would dissolve in the organic layer.
D) Both benzoic acid and NaCl would dissolve in the water layer.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the overall charge of the amino acid, alanine, at pH = 10?
A) + 1
B) - 1
C) No overall charge
D) - 2
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of increasing acidity, putting the least acidic first. 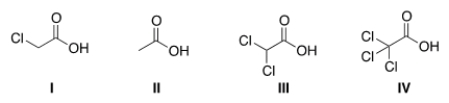
A) II < III < I < IV
B) II < I < III < IV
C) IV < III < I < II
D) IV < I < II < III
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the common name of the following compound? 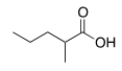
A) "-Methylbutyric acid"
B) "-Propylpropionic acid"
C) "-Methylvaleric acid"
D) "-Methylcaproic acid"
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the overall charge of the amino acid, alanine, at pH = 7?
A) + 1
B) - 1
C) No overall charge
D) + 2
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the labeled protons (Ha-Hd) in order of increasing acidity, starting with the least acidic. 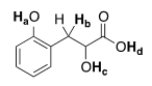
A) Ha < Hb < Hc < Hd
B) Hb < Hc < Ha < Hd
C) Hd < Ha < Hc < Hb
D) Hb < Hc < Hd < Ha
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the overall charge of the amino acid, alanine, at pH = 2?
A) + 1
B) - 1
C) No overall charge
D) + 2
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following bases are strong enough to significantly deprotonate ethanol, CH3CH2OH (pKa = 16) ?
A) NaOCH3
B) NaOH
C) NaH
D) NaOCH2CH3
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is pure acetic acid often called glacial acetic acid?
A) Because it freezes just below 0°C, forming white crystals.
B) Because it freezes just below 100°C, forming white crystals.
C) Because it freezes just below room temperature, forming white crystals.
D) Because it freezes just above room temperature, forming white crystals.
F) All of the above
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Which of the following structures is the major contributor to the resonance hybrid of the phenoxide anion? 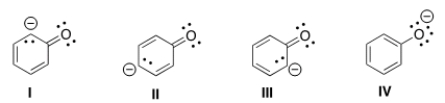
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the most polar organic compound?
A) CH3CH2CH2CH3
B) CH3CH2CHO
C) CH3CH2CH2OH
D) CH3COOH
F) B) and D)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
What is the correct IUPAC name of the following compound? 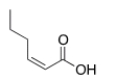
A) (E) -2-Hexenoic acid
B) (Z) -2-Hexenoic acid
C) (E) -4-Hexenoic acid
D) (Z) -4-Hexenoic acid
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 44
Related Exams