A) 1.43 µV
B) 1.43 V
C) 0.715 µV
D) 0.715 mV
E) 1.28 µV
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the classical free-electron theory of metals,
A) a metal is pictured as a regular three-dimensional array of atoms or ions.
B) a metal contains a large number of electrons that are free to move throughout the whole metal.
C) thermal equilibrium is maintained by collisions of electrons with the lattice ions.
D) electrons in the absence of an electric field move about the metal much as gas molecules move in a container.
E) All of these are true.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A semiconductor for which it is true that the major charge carriers are holes
A) is called an n-type semiconductor.
B) is called a p-type semiconductor.
C) accepts positive charges from filled valence bands.
D) is devoid of acceptor levels.
E) None of these is correct.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The crystalline structure known as hexagonal close-packed
A) is characterized by the stacking of identical spheres.
B) occurs when every ion has eight nearest-neighbor ions of opposite charge.
C) corresponds to an alternating pattern of ions.
D) occurs when an ion of either kind has six nearest neighbors of the other kind.
E) None of these is correct.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The crystalline structure known as face-centered-cubic
A) is characterized by the stacking of identical spheres.
B) occurs when every ion has eight nearest-neighbor ions of opposite charge.
C) corresponds to an alternating pattern of ions.
D) occurs when an ion of either kind has six nearest neighbors of the other kind.
E) None of these is correct.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the following to answer the question:
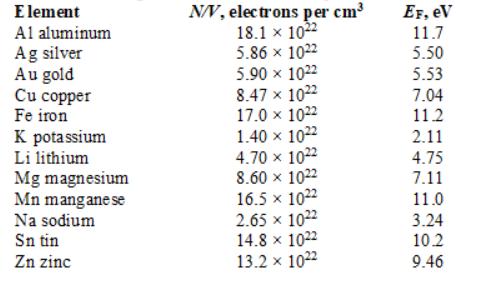
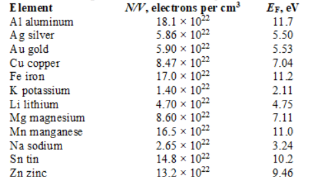 -The Fermi energy in three dimensions at T = 0 is given by
-The Fermi energy in three dimensions at T = 0 is given by 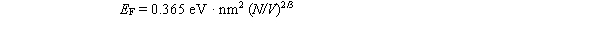 The Fermi energy for aluminum is approximately
The Fermi energy for aluminum is approximately
A) 11.7 eV
B) 3.24 eV
C) 5.53 eV
D) 2.11 eV
E) 9.46 eV
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The average energy of a Fermi electron gas at T = 0 is
A) 3kT/2
B) EF
C) kT
D) zero
E) 3EF/5
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If the energy levels of a Fermi gas is given by En = nE1,the total energy of seven fermions in the ground state is
A) 7E1
B) 9E1
C) 11E1
D) 14E1
E) 16E1
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the following to answer the question below:
 -The speed of a conduction electron whose energy is equal to the Fermi energy EF for magnesium is
-The speed of a conduction electron whose energy is equal to the Fermi energy EF for magnesium is
A) 1.42 106 m/s
B) 1.84 106 m/s
C) 1.21 106 m/s
D) 2.03 106 m/s
E) 1.58 106 m/s
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the following to answer the question. 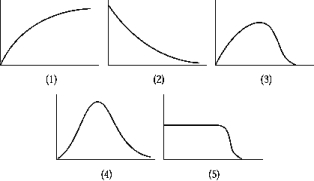 -The graph that represents the Fermi factor F is
-The graph that represents the Fermi factor F is
A) 1
B) 2
C) 3
D) 4
E) 5
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A metal is a good conductor because the valence energy band for electrons is
A) empty.
B) completely full.
C) full,but there is only a small gap to a higher empty band.
D) partly full.
E) None of these is correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The classical theory of conduction
A) fails because electrons are not classical particles.
B) does not take into account the scattering of electron waves by a lattice.
C) fails to correctly describe the collision of an electron with a lattice ion.
D) predicts a Maxwell-Boltzmann-like distribution of electron velocities.
E) is described by all of these.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The classical free-electron theory of metals
A) correctly predicts the resistivity of a conductor but fails to predict the correct temperature dependence of the resistivity.
B) correctly predicts both the resistivity of a conductor and its temperature dependence.
C) incorrectly predicts the resistivity of a conductor but correctly predicts its temperature dependence.
D) incorrectly predicts both the resistivity of a conductor and its temperature dependence.
E) correctly predicts which materials are conductors,insulators,or semiconductors.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to the BCS theory,
A) electrons in a superconductor are coupled in pairs at low temperatures.
B) electrons in a Cooper pair have equal and opposite spins.
C) electrons in a Cooper pair act as a single particle with zero spin.
D) any number of Cooper pairs may be in the same quantum state with the same energy.
E) All of these are true.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The resistivity of iron at 20ºC is 10 10-8 · m and the number of electrons per cubic centimeter is 17.0 1022.If you take the average speed of the electrons to be equal to their root-mean-square speed (at T = 300 K) of 1.17 105 m/s,it follows that their mean free path, ,must be
A) 0.245 nm
B) 0.317 nm
C) 0.428 nm
D) 0.556 nm
E) None of these is correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is false?
A) The behavior of free electrons in a metal is very different from the behavior of the molecules in an ordinary gas.
B) Ordinary gas molecules are described by the classical Maxwell-Boltzmann energy distribution function.
C) The main features of a Fermi electron gas can be understood by assuming an electron in a metal to be a particle in a box.
D) No two electrons in an atom can be in the same quantum state.
E) None of these is false.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the following to answer the question below:
 -The probability that an energy state in magnesium 0.05 eV below the Fermi energy is occupied at T = 300 K is
-The probability that an energy state in magnesium 0.05 eV below the Fermi energy is occupied at T = 300 K is
A) 5.52%
B) 20.6%
C) 47.1%
D) 87.4%
E) 99.9%
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Molecules in a liquid
A) have little influence on one another.
B) have little influence on one another except during their frequent but brief collisions.
C) are close together and exert forces on one another that are comparable to the forces that bind atoms into molecules.
D) form temporary short-range bonds that continually break and re-form due to the thermal kinetic energy of the molecules.
E) are always arranged in a crystalline structure.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If you want to produce an amorphous solid from a liquid,you
A) cool the liquid slowly so that the molecules gradually lose their kinetic energy and thus form a maximum number of bonds.
B) cool the liquid rapidly to remove its internal energy before the molecules have a chance to form many bonds.
C) maintain the temperature of the liquid while increasing the pressure applied to the liquid.
D) maintain the temperature of the liquid while decreasing the pressure applied to the liquid.
E) warm the liquid while decreasing its pressure.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The crystal structure of a substance can be determined using X-ray diffraction.It can also be determined using neutron scattering.What is the kinetic energy of a neutron that has a wavelength of 0.1 nm?
A) 0.14 eV
B) 0.082 eV
C) 0.034 eV
D) 0.23 eV
E) 0.38 eV
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 75
Related Exams