A) Carbon compounds
B) Molecular compounds
C) Ionic compounds
D) Covalent compounds
E) Ring compounds
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following general wedge-and-dash diagram:  Which of the following has this structure?
Which of the following has this structure?
A) BF3
B) CH2O
C) AlCl3
D) COCl2
E) PH3
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the best Lewis diagram for HCO2-?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following wedge-and-dash diagrams best illustrates the molecular geometry of C2H6?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is an ether?
A) C6H12
B) CH3OH
C) CH3CH2COOH
D) CH3CH2CH3
E) None is an ether.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following has a(n) bent (angular) molecular geometry?
A) OF2
B) CO2
C) H3O+
D) IO3-
E) NH4+
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecular geometries yields molecules which must be nonpolar?
A) Linear
B) Tetrahedral
C) Trigonal planar
D) All yield molecules which must be nonpolar
E) None yields molecules which must be nonpolar
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules is/are polar? (i) HF (ii) BH3 (iii) CBr2F2 (iv) PF3
A) ii only
B) i and ii
C) ii and iv
D) i,iii,and iv
E) i,ii,iii,and iv
G) A) and C)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Which of the following has a tetrahedral electron pair geometry surrounding the central atom? (i) PH3 (ii) BH3 (iii) BeH2 (iv) OF2
A) i only
B) ii only
C) iv only
D) i and ii
E) i and iv
G) A) and B)
Correct Answer

verified
E
Correct Answer
verified
Multiple Choice
What is the electron pair geometry surrounding the boron atom in BH3?
A) Trigonal planar
B) Linear
C) Angular (Bent)
D) Trigonal pyramid
E) Tetrahedral
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following best describes hydrocarbons?
A) Alkanes in which a hydrogen atom is replaced by a hydroxyl group
B) Binary compounds of carbon and hydrogen
C) Organic compounds containing water and carbon
D) Covalently bonded carbon compounds which have intermolecular force attractions to hydrogen compounds
E) Compounds which are formed by the reaction of a naturally occurring carbon-containing substance and water
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecular models has the same molecular geometry as that surrounding the oxygen atom in OF2?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following general wedge-and-dash diagrams. (i) 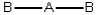 (ii)
(ii) 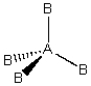 (iii)
(iii) 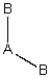 Each consists of a central atom bonded to 2 or 4 terminal atoms that are identical to one another. The central atom is different from the terminal atoms. Which of these molecules is/are polar?
Each consists of a central atom bonded to 2 or 4 terminal atoms that are identical to one another. The central atom is different from the terminal atoms. Which of these molecules is/are polar?
A) i only
B) ii only
C) iii only
D) ii and iii
E) i,ii,and iii
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular geometry surrounding the carbon atom in CH4?
A) Tetrahedral
B) Angular (bent)
C) Trigonal pyramid
D) Linear
E) Trigonal planar
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the best Lewis diagram for CH3Br?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following general wedge-and-dash diagram: 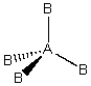 What is the molecular geometry around the central atom of this molecule?
What is the molecular geometry around the central atom of this molecule?
A) Linear
B) Tetrahedral
C) Trigonal planar
D) Bent
E) Trigonal pyramidal
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the electron pair geometry shown in the following model? 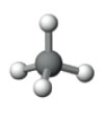
A) Tetrahedral
B) Angular (Bent)
C) Linear
D) Trigonal planar
E) Trigonal pyramid
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A molecule consists of a second period central atom with three other atoms singly bonded to it.There is one lone pair of electrons on the central atom,and the other atoms have lone pairs.What is the molecular geometry around the central atom?
A) Linear
B) Trigonal planar
C) Tetrahedral
D) Trigonal pyramid
E) Angular (bent)
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the best Lewis diagram for HCO3-?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) None of the above
Correct Answer

verified
E
Correct Answer
verified
Multiple Choice
Which of the following statements is/are correct? (i) A molecule is nonpolar if it contains only nonpolar bonds (ii) A polar molecule is one in which there is an asymmetrical distribution of charge. (iii) If the central atom in a molecule has no lone pairs and all the atoms bonded to it are identical,the molecule is nonpolar.
A) i only
B) ii only
C) iii only
D) i and iii
E) All statements are correct
G) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 47
Related Exams