A) the carbonyl group donates electrons by resonance.
B) the carbonyl group withdraws electrons by resonance.
C) the nitrogen does not have a lone pair of electrons.
D) the nitrogen has a full positive charge.
E) amides do not contain nitrogen.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The hydrolysis of esters, amides, and nitriles:
A) can be carried out under acidic or basic conditions.
B) must be acid-catalyzed.
C) must be base-catalyzed.
D) should be carried out at pH 7.0 for optimum efficiency.
E) is not pH dependent.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Cyclic amides are called:
A) lactones.
B) lactams.
C) aminals.
D) animals.
E) imines.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Essay
Provide the structure of propanoic anhydride.
Correct Answer

verified
Correct Answer
verified
Essay
How might one obtain a significant quantity of a lactone even when the formation reaction has a positive DG?
Correct Answer

verified
This reaction can be driven by...View Answer
Show Answer
Correct Answer
verified
View Answer
Essay
Predict the major product of the following reaction. 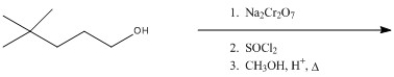
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following are strongly hydrogen bonded in the liquid phase?
A) nitriles
B) esters
C) secondary amides
D) tertiary amides
E) acid chlorides
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Provide the major organic product of the following reaction. 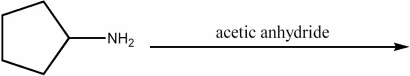
Correct Answer

verified
Correct Answer
verified
Essay
Provide the major organic product in the reaction shown below. 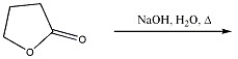
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the common name for the following carboxylic acid derivative? 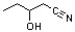
A) β-hydroxybutyronitrile
B) 1-cyano-2-butanol
C) β-hydroxyvaleronitrile
D) γ-hydroxyvaleronitrile
E) 2-hydroxypentane nitrile
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Typically, amides will hydrolyze under ________ conditions than esters.
A) stronger
B) more dilute
C) milder
D) less vigorous
E) more saline
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is(are) hydrolyzed to butanoic acid upon heating in H2O, H2SO4?
A) ethyl butanoate
B) butyl acetate
C) N-methylbutanamide
D) both A and B
E) both A and C
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Essay
Provide the major organic product in the reaction shown below. 
Correct Answer

verified
Correct Answer
verified
Essay
Using ethanol as your only source of carbon compounds and using any other necessary inorganic reagents, propose a synthesis of ethyl acetate.
Correct Answer

verified
Beginning with ethan...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
What reagent is used to convert pentanamide to 1-pentanamine?
A) POCl3
B) CuCN
C) CH3MgBr
D) SOCl2
E) LiAlH4
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Using ethanol as your only source of carbon compounds and using any other necessary inorganic reagents, propose a synthesis of acetonitrile.
Correct Answer

verified
Beginning with ethan...View Answer
Show Answer
Correct Answer
verified
View Answer
Essay
Provide the structure of o-bromobenzoyl chloride.
Correct Answer

verified
Correct Answer
verified
Essay
Using the two key resonance structures for an ester, show how resonance stabilization is lost when an ester is attacked by a nucleophile.
Correct Answer

verified
Correct Answer
verified
Essay
Predict the major product of the following reaction. 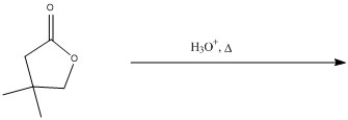
Correct Answer

verified
Correct Answer
verified
Short Answer
Provide the structure of 3-oxobutanenitrile.
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 128
Related Exams