A) Ac-234
B) Pa-234
C) Ac-235
D) Pa-235
E) Ac-233
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The 14C activity of some ancient Peruvian corn was found to be 10 disintegrations per minute per gram of carbon.If present-day plant life shows 15 dpm/g,how old is the Peruvian corn? (The half-life of 14C is 5730 yr.)
A) 1460 yr
B) 1910 yr
C) 3350 yr
D) 3820 yr
E) 9080 yr
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
In a nuclear reaction elements are converted to other elements.
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the name for the minimum mass of fissionable material required to generate a self-sustaining nuclear chain reaction?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In order to investigate the age of a mineral sample believed to be millions of years old,which would be the best choice of radioactive isotope to measure?
A) (59Fe (t1/2 = 45.1 d) )
B) (12C (t1/2 = 5.72 ×103 yr) )
C) (53Mn (t1/2 = 3.7 ×106 yr) )
D) (40K (t1/2 = 1.2 ×109 yr) )
E) (238U (t1/2 = 4.51 ×109 yr) )
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What role does cadmium metal (Cd) play in a nuclear reactor?
A) Slows down the fission neutrons (moderator)
B) Transfers heat from the reactor to the heat exchanger (primary coolant)
C) Controls chain reaction (control rods)
D) Transfers heat from the condenser to the environment (cooling tower)
E) Undergoes fission (fuel rods)
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the equation below,what particle or type of radiation needs to be included to balance the equation? 220Rn → ? + alpha particle
A) Ra-224
B) Rn-224
C) Rn-216
D) At-220
E) Po-216
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A patient's thyroid gland is to be exposed to an average of 5.5 μCi for 16 days as an ingested sample of iodine-131 decays.If the energy of the β radiation is 9.7 × 10-14 J and the mass of the thyroid is 32.0 g,what is the dose received by the patient? (1 rad = 1 × 10-5 J/g; 1 Ci = 3.7 × 1010 disintegrations/s)
A) 0.027 rads
B) 85 rads
C) 37 rads
D) 23 rads
E) None of the answers is correct.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which type of radiation is the least dangerous to humans?
A) Positrons
B) Neutrons
C) Alpha
D) Beta
E) Gamma
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which type of radiation is the most dangerous to humans?
A) Protons
B) Neutrons
C) Alpha
D) Beta
E) Gamma
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rubidium-87 decays by beta decay with a half-life of 4.9 × 1010 yr.How many 87Rb atoms are in a moon rock sample that has a rubidium decay rate of 3500 disintegrations per hour?
A) 9.0 × 1016 atoms
B) 4.3 × 10-4 atoms
C) 2.2 × 1018 atoms
D) 2.5 × 1014 atoms
E) 1.7 × 1014 atoms
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What other particle is emitted when a neutron is converted to a proton in a nucleus?
A) Gamma particle
B) Alpha particle
C) Positron
D) Beta particle
E) None of the answers is correct.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the nuclear process called where small nuclei are combined into larger ones?
A) Photonuclear reactions
B) Nuclear fission
C) Thermal conductivity
D) Nuclear combination
E) Nuclear fusion
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Cobalt-60 is a beta emitter with a half-life of 5.3 years.Approximately what fraction of cobalt-60 atoms will remain in a particular sample after 26.5 years?
A) 1/5
B) 1/16
C) 1/26
D) 1/32
E) 1/64
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
As a result of beta decay,the product nucleus is
A) one atomic number lower than the original element.
B) two atomic numbers higher than the original element.
C) one atomic number higher than the original element.
D) two atomic numbers lower than the original element.
E) four atomic numbers lower than the original element.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
Naturally occurring uranium contains approximately 1% U-235 and 99% U-238.Of these,the isotope that undergoes fission in a nuclear reactor is U-238.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is used to image the liver?
A) (18O)
B) (131I)
C) (123I)
D) (24Na)
E) (99Tc)
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the following diagrams,A represents a radioactive isotope that decays into a new isotope,X.Each circle represents 1 mmol of atoms. t = 0: 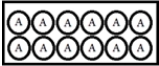 t = 25.0 s:
t = 25.0 s: 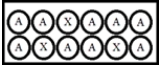 What is the half-life of A?
What is the half-life of A?
A) 6.25 s
B) 18.8 s
C) 25.0 s
D) 50.0 s
E) 60.2 s
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
The difference between the mass of an atom and the sum of the masses of its protons,neutrons,and electrons is called the ___________ ___________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is used to image the heart?
A) (18O)
B) (131I)
C) (123I)
D) (24Na)
E) (99Tc)
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Showing 101 - 120 of 125
Related Exams